Plasma Derived Therapy Market
Plasma Derived Therapy Market Size, Scope, Growth, Trends and By Segmentation Types, Applications, Regional Analysis and Industry Forecast (2025-2033)
Report ID : RI_703794 | Last Updated : August 05, 2025 |
Format : ![]()
![]()
![]()
![]()
Plasma Derived Therapy Market Size
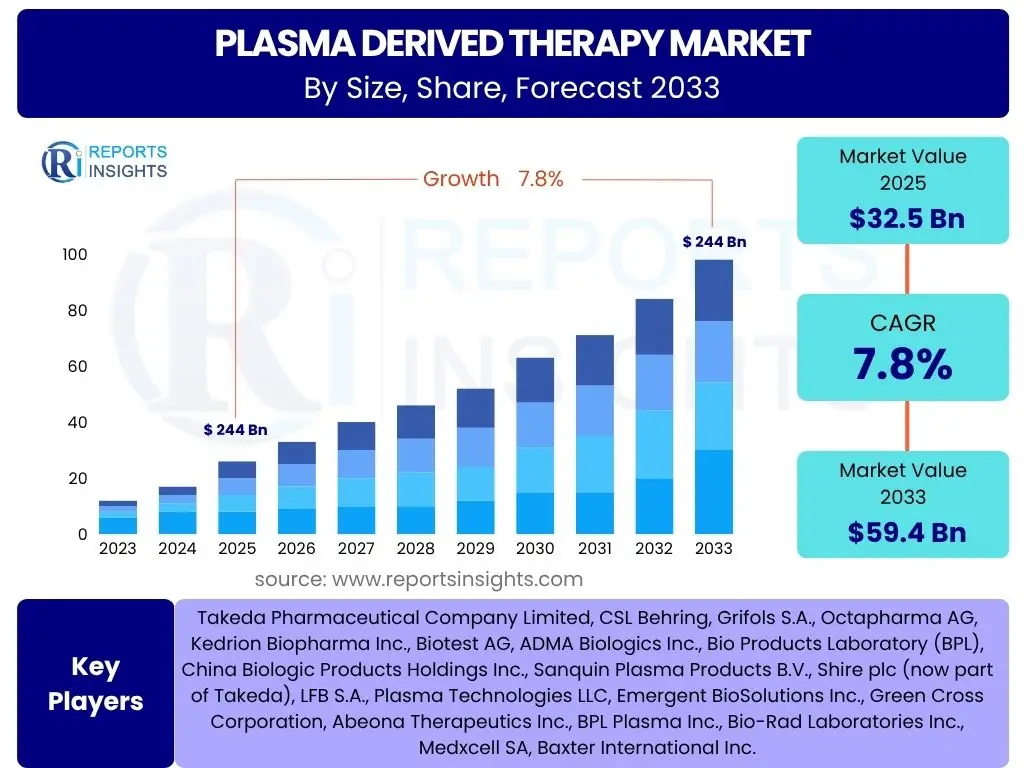
According to Reports Insights Consulting Pvt Ltd, The Plasma Derived Therapy Market is projected to grow at a Compound Annual Growth Rate (CAGR) of 7.8% between 2025 and 2033. The market is estimated at USD 32.5 Billion in 2025 and is projected to reach USD 59.4 Billion by the end of the forecast period in 2033.
Key Plasma Derived Therapy Market Trends & Insights
User inquiries frequently highlight the dynamic evolution of the Plasma Derived Therapy market, seeking information on the latest advancements, shifting demands, and the emergence of new therapeutic applications. There is a keen interest in understanding how technological innovations in plasma fractionation, purification, and diagnostic screening are influencing market growth and product safety. Furthermore, discussions often revolve around the increasing prevalence of chronic and rare diseases, which are significantly driving the demand for plasma-derived therapies, alongside the expanding geriatric population requiring advanced treatments.
Another significant area of focus for users concerns the global supply chain dynamics and the regulatory landscape impacting plasma collection and product distribution. Queries often probe into the sustainability of plasma supply, the impact of voluntary versus compensated donations, and the harmonization of international regulatory standards to ensure product availability and safety. The market is also witnessing a trend towards product diversification, with companies investing in research and development to introduce novel indications and expand the therapeutic utility of existing plasma products, thereby addressing unmet medical needs across various therapeutic areas such as immunology, neurology, and hematology.
- Increasing prevalence of rare and chronic diseases driving demand.
- Technological advancements in plasma collection, fractionation, and purification.
- Expansion of therapeutic applications for existing plasma products.
- Growing geriatric population requiring advanced immunological and neurological therapies.
- Rising awareness and improved diagnosis of primary immunodeficiencies and autoimmune disorders.
- Strategic collaborations and partnerships among market players to enhance research and market reach.
- Focus on developing recombinant protein alternatives and next-generation plasma therapies.
- Emergence of robust regulatory frameworks ensuring product safety and quality.
AI Impact Analysis on Plasma Derived Therapy
Common user questions regarding AI's impact on Plasma Derived Therapy center on its potential to revolutionize the entire value chain, from donor screening and plasma collection to product development and patient management. Users are particularly interested in how Artificial Intelligence can enhance the efficiency and safety of plasma donation centers, improve the accuracy of disease diagnosis, and optimize the production processes of plasma-derived products. There is also significant curiosity about AI's role in accelerating drug discovery for new plasma-derived treatments and in tailoring therapies for personalized medicine approaches, indicating a high expectation for transformational changes.
Furthermore, inquiries often delve into the practical applications of AI in real-world clinical settings, such as predictive analytics for patient outcomes, optimization of treatment regimens, and real-time monitoring of disease progression in patients receiving plasma therapies. Concerns about data privacy, algorithmic bias, and the ethical implications of AI in healthcare are also frequently raised. Despite these considerations, the overarching sentiment is one of optimism regarding AI's capacity to streamline operations, reduce costs, enhance product efficacy, and ultimately improve patient access to life-saving plasma-derived therapies by fostering innovation and efficiency across the industry.
- AI-driven optimization of plasma donor screening and qualification processes.
- Enhanced precision in diagnostic tools for conditions requiring plasma therapy.
- Streamlined plasma collection and logistics through predictive analytics for supply chain management.
- Acceleration of drug discovery and development for novel plasma-derived products.
- Personalized treatment recommendations based on patient data analysis by AI algorithms.
- Improved quality control and safety monitoring in plasma fractionation and manufacturing.
- Predictive modeling for patient response to plasma therapies, optimizing dosage and outcomes.
- Automation of administrative and repetitive tasks within plasma centers, increasing efficiency.
Key Takeaways Plasma Derived Therapy Market Size & Forecast
Analysis of common user questions regarding the Plasma Derived Therapy market size and forecast reveals a primary interest in understanding the core growth drivers and the long-term sustainability of this sector. Users frequently inquire about the specific factors contributing to the projected CAGR, such as disease prevalence, healthcare infrastructure development, and policy changes. There is a strong focus on identifying which product segments and geographic regions are expected to exhibit the most significant growth, signaling investment interest and strategic planning opportunities within the market.
Furthermore, users often seek clarity on the potential impact of emerging technologies, competitive landscapes, and regulatory shifts on the market's trajectory towards its forecasted valuation. The insights derived suggest that the market is poised for sustained expansion, driven by continuous innovation in therapeutic applications and an increasing global burden of target diseases. The forecast indicates robust demand for essential plasma-derived products, positioning the market as a critical and expanding segment within the biopharmaceutical industry, with significant opportunities for both established players and new entrants who can address supply chain efficiencies and therapeutic diversification.
- Robust market growth driven by increasing incidence of chronic and rare diseases globally.
- Significant expansion projected across immunoglobulin and albumin product segments.
- North America and Europe to remain dominant markets, while Asia Pacific exhibits fastest growth.
- Technological advancements in purification and fractionation are key enablers of market expansion.
- Strategic investments in research and development for new indications will bolster future growth.
- Supply chain resilience and plasma donor recruitment remain critical success factors.
- The market offers substantial opportunities for innovation and geographic expansion.
Plasma Derived Therapy Market Drivers Analysis
The Plasma Derived Therapy market is significantly propelled by several intrinsic and extrinsic factors that collectively contribute to its robust growth trajectory. A primary driver is the escalating global prevalence of chronic and rare diseases, including various immunodeficiencies, neurological disorders, and bleeding disorders, which necessitate the use of plasma-derived products for effective management and treatment. Alongside this, advancements in diagnostic capabilities have led to improved identification of these conditions, thereby expanding the patient pool requiring therapy.
Moreover, the continuous innovation in plasma fractionation technologies and the development of new therapeutic applications for existing plasma products are fueling market expansion. This includes the exploration of plasma derivatives for conditions previously untreated or inadequately managed. Additionally, the growing geriatric population, which is more susceptible to age-related illnesses requiring plasma-based interventions, coupled with increasing healthcare expenditure and improving access to specialized treatments in emerging economies, further accelerates market demand.
| Drivers | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Increasing Prevalence of Rare & Chronic Diseases | +1.5% | Global, particularly North America, Europe | Long-term (2025-2033) |
| Advancements in Plasma Fractionation Technologies | +0.8% | Developed Regions (US, EU, Japan) | Mid-term (2027-2031) |
| Growing Geriatric Population | +0.7% | Global, notably Europe, North America, East Asia | Long-term (2025-2033) |
| Rising Awareness & Diagnosis of Immunodeficiencies | +0.6% | Global, especially emerging economies | Mid-term (2026-2030) |
| Expanding Therapeutic Applications | +0.9% | Global | Long-term (2025-2033) |
Plasma Derived Therapy Market Restraints Analysis
Despite the positive growth outlook, the Plasma Derived Therapy market faces several significant restraints that could impede its expansion. One of the primary limitations is the inherently high cost associated with the production and administration of plasma-derived products, making them less accessible in price-sensitive markets or for patients without robust insurance coverage. This economic barrier can restrict market penetration and limit patient access, particularly in developing regions.
Furthermore, the market is constrained by stringent regulatory frameworks and complex approval processes imposed by health authorities worldwide. These regulations, while crucial for ensuring product safety and efficacy, often lead to prolonged development timelines and increased R&D costs, thereby impacting the speed of new product introduction. Concerns regarding the risk of pathogen transmission, though significantly mitigated by advanced screening and purification technologies, also remain a public health apprehension that can influence patient and physician acceptance. Lastly, the dependency on a consistent and adequate supply of human plasma, which relies on donor recruitment and retention efforts, poses a perennial challenge to meeting global demand.
| Restraints | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| High Cost of Plasma-Derived Therapies | -0.9% | Global, particularly developing economies | Long-term (2025-2033) |
| Stringent Regulatory Approval Processes | -0.7% | Global | Long-term (2025-2033) |
| Risk of Pathogen Transmission (perceived/actual) | -0.5% | Global | Mid-term (2026-2030) |
| Limited Plasma Collection Infrastructure | -0.6% | Emerging Markets | Long-term (2025-2033) |
| Ethical Concerns Regarding Plasma Donation | -0.3% | Europe, North America | Long-term (2025-2033) |
Plasma Derived Therapy Market Opportunities Analysis
The Plasma Derived Therapy market presents a myriad of growth opportunities driven by evolving healthcare needs and technological progress. A significant opportunity lies in the development of novel indications for existing plasma products, expanding their therapeutic utility beyond traditional applications into areas like neurology, oncology, and critical care. This continuous exploration of new therapeutic avenues allows for market diversification and addresses a wider spectrum of unmet medical needs, thereby unlocking new revenue streams for manufacturers.
Furthermore, geographic expansion into emerging markets such as Asia Pacific, Latin America, and the Middle East and Africa offers substantial growth prospects. These regions are characterized by improving healthcare infrastructure, rising disposable incomes, and increasing awareness of plasma-derived therapies, representing largely untapped markets for current and future products. Technological innovations in plasma purification, advent of recombinant alternatives, and advanced diagnostic methods also create opportunities to enhance product safety, efficacy, and production efficiency, potentially reducing costs and improving patient access globally.
| Opportunities | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Development of Novel Therapeutic Indications | +1.0% | Global | Long-term (2025-2033) |
| Expansion into Emerging Markets | +1.2% | Asia Pacific, Latin America, MEA | Long-term (2025-2033) |
| Technological Innovations in Purification Methods | +0.7% | Developed Regions | Mid-term (2027-2031) |
| Strategic Collaborations and Acquisitions | +0.5% | Global | Mid-term (2026-2030) |
| Growing Demand for Specialty Plasma Products | +0.6% | North America, Europe | Long-term (2025-2033) |
Plasma Derived Therapy Market Challenges Impact Analysis
The Plasma Derived Therapy market faces persistent challenges that necessitate strategic responses from industry participants. One significant challenge is ensuring a stable and sufficient supply of source plasma to meet the ever-increasing global demand for life-saving therapies. This involves overcoming logistical hurdles in plasma collection, managing donor recruitment and retention, and navigating potential supply chain disruptions, which can directly impact product availability and market stability.
Another crucial challenge is intense competition from alternative therapies, including recombinant proteins and gene therapies, which offer potential advantages in terms of supply consistency and reduced risk of pathogen transmission. This competitive landscape mandates continuous innovation and differentiation for plasma-derived products. Furthermore, the high research and development costs associated with bringing new plasma therapies to market, coupled with the need to adhere to complex and evolving regulatory standards across diverse jurisdictions, add significant financial and operational burdens on manufacturers, potentially delaying market entry and limiting investment in certain areas.
| Challenges | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Ensuring Consistent Plasma Supply | -0.8% | Global | Long-term (2025-2033) |
| Competition from Alternative Therapies | -0.6% | Global | Long-term (2025-2033) |
| High Research & Development Costs | -0.4% | Global | Long-term (2025-2033) |
| Regulatory Complexity Across Regions | -0.5% | Global | Long-term (2025-2033) |
| Public Perception and Donor Engagement | -0.3% | Developed Regions | Mid-term (2026-2030) |
Plasma Derived Therapy Market - Updated Report Scope
This comprehensive report provides an in-depth analysis of the Plasma Derived Therapy market, encompassing historical data, current market dynamics, and future projections. It delivers critical insights into market size, growth drivers, restraints, opportunities, and challenges across various segments and key geographical regions. The scope also includes a detailed competitive landscape, profiling leading market players and their strategic initiatives, alongside an examination of emerging trends and their potential impact on the industry's evolution.
| Report Attributes | Report Details |
|---|---|
| Base Year | 2024 |
| Historical Year | 2019 to 2023 |
| Forecast Year | 2025 - 2033 |
| Market Size in 2025 | USD 32.5 Billion |
| Market Forecast in 2033 | USD 59.4 Billion |
| Growth Rate | 7.8% |
| Number of Pages | 247 |
| Key Trends |
|
| Segments Covered |
|
| Key Companies Covered | Takeda Pharmaceutical Company Limited, CSL Behring, Grifols S.A., Octapharma AG, Kedrion Biopharma Inc., Biotest AG, ADMA Biologics Inc., Bio Products Laboratory (BPL), China Biologic Products Holdings Inc., Sanquin Plasma Products B.V., Shire plc (now part of Takeda), LFB S.A., Plasma Technologies LLC, Emergent BioSolutions Inc., Green Cross Corporation, Abeona Therapeutics Inc., BPL Plasma Inc., Bio-Rad Laboratories Inc., Medxcell SA, Baxter International Inc. |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Speak to Analyst | Avail customised purchase options to meet your exact research needs. Request For Analyst Or Customization |
Segmentation Analysis
The Plasma Derived Therapy market is broadly segmented based on product type, application, and end-use, providing a granular view of market dynamics and growth opportunities within each category. This segmentation helps in understanding the specific demand drivers and therapeutic requirements across different patient populations and healthcare settings. Each segment plays a crucial role in the overall market landscape, reflecting diverse medical needs and the targeted development of plasma-derived solutions.
Product segmentation highlights the dominance of immunoglobulins due to their wide application in immunodeficiencies and autoimmune disorders, followed by albumin and coagulation factors critical for blood disorders. Application segmentation underscores the significant use in immunology, neurology, and hematology, reflecting the burden of diseases within these therapeutic areas. The end-use segment demonstrates the primary channels through which these therapies are administered, with hospitals and clinics being the major consumers, complemented by a growing role for specialty pharmacies and research institutions.
- By Product:
- Immunoglobulins
- Intravenous Immunoglobulin (IVIG)
- Subcutaneous Immunoglobulin (SCIG)
- Albumin
- Coagulation Factors
- Factor VIII
- Factor IX
- Von Willebrand Factor
- Hyperimmune Globulins
- Hepatitis B Immune Globulin
- Tetanus Immune Globulin
- Rabies Immune Globulin
- Rho(D) Immune Globulin
- Others
- Alpha-1 Antitrypsin
- Others (Protein C, C1 Esterase Inhibitor, etc.)
- Immunoglobulins
- By Application:
- Immunology
- Primary Immunodeficiency
- Secondary Immunodeficiency
- Autoimmune Diseases
- Neurology
- Chronic Inflammatory Demyelinating Polyneuropathy (CIDP)
- Multifocal Motor Neuropathy (MMN)
- Hematology
- Hemophilia A
- Hemophilia B
- Von Willebrand Disease
- Pulmonology
- Alpha-1 Antitrypsin Deficiency
- Critical Care
- Others (Hereditary Angioedema, etc.)
- Immunology
- By End-use:
- Hospitals & Clinics
- Academic & Research Institutes
- Specialty Pharmacies
Regional Highlights
- North America: This region is anticipated to hold the largest market share due to its well-established healthcare infrastructure, high prevalence of target diseases, significant R&D investments in biopharmaceuticals, and robust reimbursement policies. The U.S. leads the market with a strong presence of key manufacturers and extensive plasma collection networks.
- Europe: Europe represents a substantial market share, driven by a growing aging population, increasing diagnosis of immunodeficiencies, and favorable government initiatives supporting plasma donation and therapy development. Countries like Germany, France, and the UK are key contributors to the regional market growth.
- Asia Pacific (APAC): Expected to be the fastest-growing region, APAC offers significant growth opportunities owing to improving healthcare infrastructure, rising disposable incomes, increasing awareness about rare diseases, and a large patient base. Countries such as China, Japan, and India are rapidly expanding their capacities for plasma collection and processing.
- Latin America: This region exhibits steady growth, primarily fueled by increasing healthcare spending, improving access to advanced treatments, and a rising prevalence of chronic diseases. Brazil and Mexico are emerging as important markets within this region.
- Middle East and Africa (MEA): The MEA region is projected to witness moderate growth, driven by increasing investments in healthcare facilities, growing awareness of plasma-derived therapies, and efforts to improve the diagnosis and treatment of inherited blood disorders and immunodeficiencies.
Top Key Players
The market research report includes a detailed profile of leading stakeholders in the Plasma Derived Therapy Market.- Takeda Pharmaceutical Company Limited
- CSL Behring
- Grifols S.A.
- Octapharma AG
- Kedrion Biopharma Inc.
- Biotest AG
- ADMA Biologics Inc.
- Bio Products Laboratory (BPL)
- China Biologic Products Holdings Inc.
- Sanquin Plasma Products B.V.
- LFB S.A.
- Plasma Technologies LLC
- Emergent BioSolutions Inc.
- Green Cross Corporation
- Abeona Therapeutics Inc.
- BPL Plasma Inc.
- Bio-Rad Laboratories Inc.
- Medxcell SA
- Baxter International Inc.
- AstraZeneca plc (for Shire plc integration)
Frequently Asked Questions
What are plasma derived therapies?
Plasma derived therapies are biopharmaceutical products made from human blood plasma. They are used to treat a wide range of serious and rare diseases, including immune deficiencies, bleeding disorders, and neurological conditions, by replacing missing or deficient proteins in the body.
What diseases can plasma derived therapies treat?
These therapies are primarily used for conditions such as primary and secondary immunodeficiencies, autoimmune disorders (e.g., CIDP), bleeding disorders (e.g., hemophilia A and B), alpha-1 antitrypsin deficiency, and various neurological conditions, among others.
How is plasma collected for these therapies?
Plasma is collected through a process called plasmapheresis, where blood is drawn from a donor, plasma is separated, and the remaining blood components are returned to the donor. This process allows for more frequent donations of plasma compared to whole blood.
What are the primary challenges in the plasma derived therapy market?
Key challenges include ensuring a stable and sufficient supply of source plasma, managing the high costs associated with production, navigating complex global regulatory environments, and facing competition from alternative or recombinant therapies.
What is the growth outlook for the plasma derived therapy market?
The plasma derived therapy market is projected for robust growth, driven by increasing prevalence of target diseases, ongoing advancements in plasma processing technologies, expansion into new therapeutic indications, and growing healthcare access in emerging economies.

