Pharmaceutical Marijuana Market
Pharmaceutical Marijuana Market Size, Scope, Growth, Trends and By Segmentation Types, Applications, Regional Analysis and Industry Forecast (2025-2033)
Report ID : RI_702832 | Last Updated : August 01, 2025 |
Format : ![]()
![]()
![]()
![]()
Pharmaceutical Marijuana Market Size
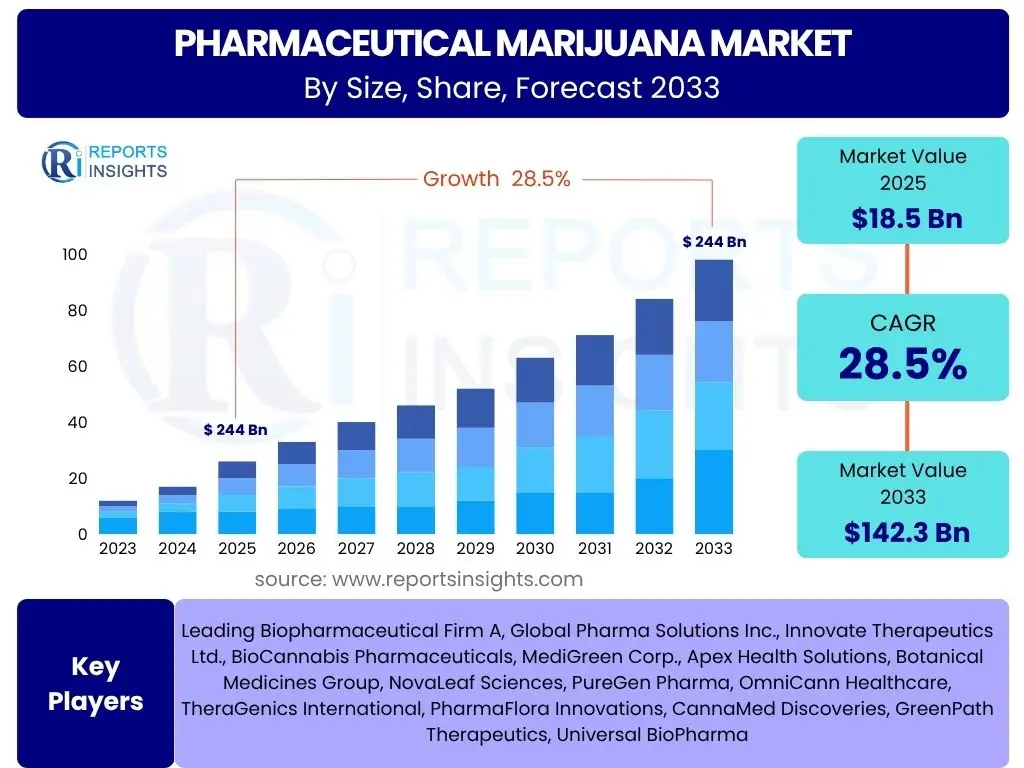
According to Reports Insights Consulting Pvt Ltd, The Pharmaceutical Marijuana Market is projected to grow at a Compound Annual Growth Rate (CAGR) of 28.5% between 2025 and 2033. The market is estimated at USD 18.5 Billion in 2025 and is projected to reach USD 142.3 Billion by the end of the forecast period in 2033.
Key Pharmaceutical Marijuana Market Trends & Insights
User queries frequently highlight the evolving landscape of pharmaceutical marijuana, focusing on how regulatory shifts are influencing market expansion and the development of new medical applications. There is considerable interest in understanding the ongoing research and development efforts, particularly in synthesizing specific cannabinoids and exploring novel delivery methods. Questions also arise regarding the increasing integration of cannabis-based therapies into conventional healthcare systems and the growing acceptance among medical professionals and patients. The market is witnessing a significant drive towards standardized dosages and formulations, moving away from anecdotal evidence to evidence-based medicine.
Another key area of user inquiry revolves around the regional disparities in market growth, driven by varying legal frameworks and healthcare infrastructures. The trend towards personalized medicine, where specific cannabinoid profiles are tailored to individual patient needs, is also a prominent topic. Furthermore, users are keen on understanding the role of technological advancements in cultivation, extraction, and product formulation, which are crucial for ensuring product quality, consistency, and safety within the pharmaceutical sector. This continuous innovation is propelling the market forward, attracting significant investments and fostering a competitive environment.
- Increasing global acceptance and legalization for medical use.
- Shift towards evidence-based research and clinical trials for new indications.
- Development of advanced drug delivery systems (e.g., inhalers, patches, sublingual films).
- Rising focus on specific cannabinoid isolation and synthetic production for targeted therapies.
- Integration of cannabis-derived medicines into mainstream healthcare.
- Growing demand for standardized and pharmaceutical-grade products.
AI Impact Analysis on Pharmaceutical Marijuana
Common user questions related to the impact of AI on pharmaceutical marijuana often center on its potential to revolutionize drug discovery, personalize treatment regimens, and optimize complex supply chains. Users are curious about how artificial intelligence can accelerate the identification of novel therapeutic compounds from cannabis, predict patient responses to different cannabinoid profiles, and improve the efficiency of clinical trials. The ethical implications of AI in healthcare, particularly concerning data privacy and algorithmic bias in patient selection, are also significant themes of inquiry. There is an expectation that AI will enhance the precision and safety of cannabis-based medicines, moving the industry towards more data-driven and patient-centric approaches.
Furthermore, users frequently inquire about AI's role in improving the cultivation and processing of pharmaceutical-grade cannabis, from optimizing growth conditions to ensuring quality control and consistency. The application of AI in managing vast datasets related to patient outcomes, cannabinoid interactions, and genetic variations is seen as critical for unlocking the full therapeutic potential of cannabis. While there is general optimism about AI's transformative capabilities, concerns about regulatory frameworks catching up with technological advancements and the need for robust validation of AI-driven insights are also prevalent. The consensus is that AI will be a cornerstone in the future development and deployment of pharmaceutical marijuana, streamlining operations and enhancing therapeutic efficacy.
- Accelerating drug discovery and development of cannabinoid-based therapies.
- Enabling personalized medicine through predictive analytics for patient response and dosing.
- Optimizing cultivation conditions for cannabinoid yield and consistency.
- Enhancing quality control and ensuring product safety through advanced data analysis.
- Streamlining supply chain management and logistics from cultivation to distribution.
- Improving clinical trial design and patient stratification for more efficient research.
Key Takeaways Pharmaceutical Marijuana Market Size & Forecast
User inquiries concerning key takeaways from the pharmaceutical marijuana market size and forecast consistently highlight the market's robust growth trajectory and its significant potential for investment. There is a strong interest in understanding the primary catalysts driving this expansion, such as evolving legal landscapes and increasing medical acceptance, alongside the critical role of ongoing research and development in unlocking new therapeutic applications. Users also seek clarity on the long-term sustainability of this growth, considering potential regulatory hurdles and shifts in public perception. The insights gathered emphasize the transition of cannabis from a recreational substance to a legitimate pharmaceutical product, driven by scientific validation and clinical efficacy.
Furthermore, the questions reveal a focus on identifying lucrative segments within the market, such as specific product types or therapeutic areas that are poised for rapid adoption. The importance of strategic partnerships, technological innovation in production, and effective distribution channels are also frequently underscored. The market forecast indicates a sustained period of expansion, underpinned by increasing healthcare expenditure, a rising prevalence of chronic conditions, and continuous advancements in cannabis-derived drug formulations. These elements collectively paint a picture of a dynamic market with considerable upside potential, contingent on navigating complex regulatory and scientific challenges.
- The pharmaceutical marijuana market exhibits strong growth potential, driven by global medical legalization.
- Significant investment opportunities exist across research, cultivation, and product development.
- Regulatory frameworks and clinical validation are pivotal for sustained market expansion.
- Innovation in cannabinoid isolation and delivery systems will be key differentiators.
- The market is transitioning towards a science-backed, pharmaceutical-grade product landscape.
- Strategic alliances and technological advancements are crucial for competitive advantage.
Pharmaceutical Marijuana Market Drivers Analysis
The pharmaceutical marijuana market is experiencing substantial growth propelled by several key drivers. Foremost among these is the escalating global trend of medical cannabis legalization and decriminalization, which has significantly expanded patient access and reduced previous barriers to entry. This legislative shift is often accompanied by increasing public and medical community acceptance of cannabis as a legitimate therapeutic agent, spurred by growing scientific evidence supporting its efficacy in managing various chronic and debilitating conditions. The rising prevalence of these conditions, such as chronic pain, neurological disorders, and certain types of cancer, further amplifies the demand for alternative or complementary treatment options.
Additionally, significant investments in research and development aimed at exploring the full therapeutic potential of cannabinoids are driving innovation, leading to the discovery of new applications and the development of standardized, pharmaceutical-grade products. Pharmaceutical companies are increasingly interested in cannabis-derived medicines, fostering collaborations and strategic partnerships that bring rigorous clinical trials and advanced drug formulation techniques to the forefront. The growing awareness among patients and healthcare providers about the potential benefits of pharmaceutical marijuana, coupled with an increasing number of clinical studies validating its safety and efficacy, are collectively contributing to the market's upward trajectory, making it a compelling area for growth and development.
| Drivers | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Increasing Global Legalization & Medical Acceptance | +8.5% | North America, Europe, Australia, Latin America | Short to Mid-term (2025-2030) |
| Rising Prevalence of Chronic Diseases | +7.0% | Globally, particularly developed economies | Mid to Long-term (2027-2033) |
| Growing Investment in R&D and Clinical Trials | +6.0% | North America, Europe, Israel | Short to Mid-term (2025-2030) |
| Demand for Alternative Pain Management & Mental Health Solutions | +4.5% | Globally, especially high-income countries | Short to Mid-term (2025-2030) |
| Advancements in Cultivation & Extraction Technologies | +2.5% | Globally, key cultivation hubs | Mid-term (2026-2031) |
Pharmaceutical Marijuana Market Restraints Analysis
Despite its significant growth potential, the pharmaceutical marijuana market faces several notable restraints that could temper its expansion. A primary challenge stems from the complex and often inconsistent regulatory frameworks across different countries and even within regions of the same country. While some jurisdictions have legalized medical cannabis, others maintain strict prohibitions, creating a fragmented global market and hindering international trade and research collaboration. This regulatory ambiguity can lead to significant compliance costs, market access barriers, and uncertainty for businesses operating in this space. The evolving nature of these laws also poses an ongoing challenge for companies trying to adapt their strategies.
Another significant restraint is the lingering social stigma associated with cannabis use, even in a medical context. Despite increasing awareness of its therapeutic benefits, historical perceptions and misconceptions continue to influence public opinion and may deter some patients and healthcare providers from considering pharmaceutical marijuana as a viable treatment option. Furthermore, the high cost of research and development, particularly for conducting rigorous clinical trials to meet pharmaceutical standards, can be prohibitive for many companies. Competition from the illicit cannabis market, which often offers lower prices due to a lack of regulation and taxation, also presents a challenge, as it can divert potential consumers away from regulated pharmaceutical products. These combined factors necessitate careful navigation for market participants.
| Restraints | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Strict & Varying Regulatory Frameworks | -5.0% | Globally, particularly Asia, parts of Europe | Short to Long-term (2025-2033) |
| Lingering Social Stigma & Public Perception | -3.5% | Globally, more pronounced in conservative regions | Mid-term (2027-2032) |
| High R&D and Clinical Trial Costs | -2.0% | Globally, especially for smaller companies | Long-term (2028-2033) |
| Competition from Illicit Market | -1.5% | Regions with newly legalized markets | Short-term (2025-2028) |
| Lack of Standardization & Quality Control Challenges | -1.0% | Emerging markets, less regulated areas | Short to Mid-term (2025-2030) |
Pharmaceutical Marijuana Market Opportunities Analysis
The pharmaceutical marijuana market is replete with significant opportunities driven by ongoing scientific advancements and evolving market dynamics. One major area of opportunity lies in the expansion of therapeutic applications beyond commonly accepted uses. As research delves deeper into the endocannabinoid system, new indications for cannabis-based medicines in areas such as neurodegenerative diseases, autoimmune disorders, and mental health conditions are continually being identified, opening up vast untapped markets. The development of novel product formulations, including highly precise dosage forms, advanced delivery systems like transdermal patches or metered-dose inhalers, and targeted cannabinoid profiles, presents a strong avenue for market differentiation and value creation, catering to specific patient needs and preferences.
Furthermore, emerging markets in Asia Pacific, Latin America, and parts of Africa, where medical cannabis laws are just beginning to take shape, represent substantial long-term growth prospects. These regions often have large patient populations and increasing healthcare expenditures, making them attractive for early market entrants. Strategic partnerships between pharmaceutical companies, cannabis cultivators, and research institutions can leverage complementary expertise, accelerate product development, and facilitate market penetration. Additionally, the increasing focus on personalized medicine and nutraceutical integration offers avenues for product diversification and consumer engagement. The continuous advancement in cultivation and extraction technologies also promises to improve product purity, potency, and cost-efficiency, further bolstering market opportunities and ensuring a stable supply of pharmaceutical-grade raw materials.
| Opportunities | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Expansion into New Therapeutic Indications | +7.0% | Globally, particularly high R&D regions | Mid to Long-term (2027-2033) |
| Development of Novel Formulations & Delivery Systems | +6.5% | North America, Europe, Australia | Short to Mid-term (2025-2030) |
| Emerging Markets in Asia Pacific & Latin America | +5.0% | China, India, Brazil, Mexico | Mid to Long-term (2028-2033) |
| Strategic Partnerships & Collaborations | +4.0% | Globally, cross-industry | Short to Mid-term (2025-2030) |
| Technological Innovation in Cultivation & Processing | +3.0% | Globally | Short to Mid-term (2025-2030) |
Pharmaceutical Marijuana Market Challenges Impact Analysis
The pharmaceutical marijuana market, while promising, faces several inherent challenges that demand strategic navigation. One significant hurdle is the persistent divergence in international drug control treaties and national laws, which creates a complex legal patchwork, impeding seamless cross-border trade, research, and product development. This regulatory fragmentation can lead to compliance complexities and inhibit the establishment of global supply chains. Furthermore, ensuring consistent quality and standardization of pharmaceutical-grade cannabis products remains a formidable challenge. Variability in plant genetics, cultivation practices, and extraction methods can lead to inconsistent cannabinoid profiles, affecting therapeutic efficacy and patient safety, and making it difficult to meet rigorous pharmaceutical manufacturing standards.
Another critical challenge is the limited long-term clinical research data available, particularly regarding the efficacy and safety of specific cannabinoids for a wide range of medical conditions. The historical prohibition of cannabis has constrained extensive large-scale clinical trials, resulting in a knowledge gap that needs to be addressed to gain broader acceptance among mainstream medical professionals and regulatory bodies. Intellectual property rights for cannabis-based innovations are also complex, given the plant's natural origin and varied genetic expressions, leading to difficulties in patenting and protecting novel discoveries. Finally, maintaining positive public perception and overcoming historical stigmas associated with cannabis use, even for medical purposes, continues to be a societal challenge, influencing patient adoption rates and policy decisions. Addressing these challenges is paramount for the sustained growth and maturity of the pharmaceutical marijuana market.
| Challenges | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Varying International Drug Control Laws & Regulations | -4.5% | Globally, cross-border trade | Long-term (2025-2033) |
| Quality Control & Standardization Issues | -3.0% | Globally, especially in new production regions | Short to Mid-term (2025-2030) |
| Limited Long-Term Clinical Research Data | -2.5% | Globally, for widespread medical acceptance | Long-term (2028-2033) |
| Intellectual Property Rights Protection | -1.5% | North America, Europe | Mid-term (2027-2032) |
| Public & Physician Education and Acceptance | -1.0% | Globally | Short to Mid-term (2025-2030) |
Pharmaceutical Marijuana Market - Updated Report Scope
This report provides an in-depth analysis of the global Pharmaceutical Marijuana Market, offering a comprehensive overview of market size, trends, drivers, restraints, opportunities, and challenges. It delves into the impact of emerging technologies like Artificial Intelligence on market dynamics and outlines key takeaways from market forecasts. The report also details market segmentation by various parameters, providing granular insights into product types, applications, routes of administration, sources, and distribution channels. Regional highlights are included to illustrate market variations and growth potentials across key geographies. Finally, the report profiles leading market players, offering a holistic view of the competitive landscape and strategic initiatives shaping the industry's future.
| Report Attributes | Report Details |
|---|---|
| Base Year | 2024 |
| Historical Year | 2019 to 2023 |
| Forecast Year | 2025 - 2033 |
| Market Size in 2025 | USD 18.5 Billion |
| Market Forecast in 2033 | USD 142.3 Billion |
| Growth Rate | 28.5% |
| Number of Pages | 250 |
| Key Trends |
|
| Segments Covered |
|
| Key Companies Covered | Leading Biopharmaceutical Firm A, Global Pharma Solutions Inc., Innovate Therapeutics Ltd., BioCannabis Pharmaceuticals, MediGreen Corp., Apex Health Solutions, Botanical Medicines Group, NovaLeaf Sciences, PureGen Pharma, OmniCann Healthcare, TheraGenics International, PharmaFlora Innovations, CannaMed Discoveries, GreenPath Therapeutics, Universal BioPharma |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Speak to Analyst | Avail customised purchase options to meet your exact research needs. Request For Analyst Or Customization |
Segmentation Analysis
The pharmaceutical marijuana market is segmented to provide a granular understanding of its diverse components, offering insights into various product categories, therapeutic applications, administration methods, and distribution channels. This detailed breakdown helps in identifying niche markets, understanding consumer preferences, and assessing the competitive landscape within specific sub-segments. By analyzing these segments, stakeholders can pinpoint high-growth areas, allocate resources effectively, and tailor product development and marketing strategies to meet specific market demands. The segmentation also highlights the evolving nature of the industry as new product forms and therapeutic uses emerge.
Understanding these segmentations is crucial for strategic planning. For instance, the distinction between CBD and THC dominance, or the emergence of other cannabinoids like CBG and CBN, indicates a trend towards more targeted therapies. Similarly, analyzing applications such as chronic pain versus neurological disorders reveals the breadth of medical conditions that pharmaceutical marijuana addresses. The growth across various routes of administration, from traditional oral forms to innovative transdermal patches, reflects advancements in drug delivery. Furthermore, the varying prominence of distribution channels, including hospital pharmacies and online platforms, underscores shifts in patient access and purchasing behaviors. This comprehensive segmentation framework is vital for comprehending the multifaceted nature of the pharmaceutical marijuana market and identifying future opportunities.
- By Product Type:
- Cannabidiol (CBD)
- Tetrahydrocannabinol (THC)
- Balanced CBD & THC
- Other Cannabinoids (e.g., Cannabigerol (CBG), Cannabinol (CBN), Cannabichromene (CBC))
- By Application:
- Chronic Pain Management
- Neurological Disorders
- Epilepsy
- Multiple Sclerosis
- Parkinson's Disease
- Other Neurological Conditions
- Cancer Management (Pain, Nausea, Appetite Stimulation)
- Mental Health Disorders (Anxiety, Depression, PTSD)
- Autoimmune Diseases (e.g., Crohn's Disease, Rheumatoid Arthritis)
- Other Applications (e.g., Glaucoma, Sleep Disorders, Appetite Stimulation for HIV/AIDS)
- By Route of Administration:
- Oral (Capsules, Tablets, Edibles, Tinctures, Syrups)
- Inhalation (Vaporizers, Sprays)
- Topical (Creams, Gels, Patches, Ointments)
- Sublingual
- Other Routes (e.g., Suppositories, Injectables)
- By Source:
- Natural (Plant-derived)
- Synthetic
- By Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Specialty Clinics & Dispensaries
Regional Highlights
- North America: This region dominates the pharmaceutical marijuana market, primarily driven by the progressive legalization of medical cannabis in the United States and Canada. High patient awareness, significant investment in R&D, and the presence of numerous key market players contribute to its leading position. The robust regulatory frameworks in some states and provinces provide a structured environment for pharmaceutical development and commercialization.
- Europe: Europe represents a rapidly growing market, fueled by increasing acceptance of medical cannabis in countries like Germany, the UK, and Italy. Regulatory reforms are gradually expanding patient access, and there's a strong focus on scientific research and the development of pharmaceutical-grade products. Demand is particularly high for cannabinoid-based drugs for neurological and pain management indications.
- Asia Pacific (APAC): The APAC region is an emerging market with substantial long-term potential. While regulatory environments remain stringent in many countries, increasing awareness and the high prevalence of chronic diseases are slowly pushing for medical cannabis legalization. Countries like Australia and South Korea are at the forefront, with others showing signs of potential future growth as legal frameworks evolve.
- Latin America: This region is experiencing considerable growth due to increasing medical cannabis legalization efforts in countries such as Colombia, Uruguay, and Mexico. Favorable cultivation conditions and a growing patient base seeking alternative therapies contribute to its market expansion. The focus is often on exporting raw materials and developing affordable pharmaceutical products.
- Middle East and Africa (MEA): The MEA market is currently nascent but holds future promise. Israel is a global leader in cannabis research and development, influencing regulatory discussions in other parts of the region. As healthcare infrastructure improves and medical acceptance grows, specific countries within MEA may emerge as significant contributors to the pharmaceutical marijuana market.
Top Key Players
The market research report includes a detailed profile of leading stakeholders in the Pharmaceutical Marijuana Market.- Leading Biopharmaceutical Firm A
- Global Pharma Solutions Inc.
- Innovate Therapeutics Ltd.
- BioCannabis Pharmaceuticals
- MediGreen Corp.
- Apex Health Solutions
- Botanical Medicines Group
- NovaLeaf Sciences
- PureGen Pharma
- OmniCann Healthcare
- TheraGenics International
- PharmaFlora Innovations
- CannaMed Discoveries
- GreenPath Therapeutics
- Universal BioPharma
- Wellness Cannabinoid Systems
- Clinical Cannabis Research
- Integrated Bio-Therapeutics
- Botanical Drug Innovators
- Precision Cannabinoid Labs
Frequently Asked Questions
What is pharmaceutical marijuana?
Pharmaceutical marijuana refers to cannabis-derived products, including specific cannabinoids like CBD or THC, that are manufactured and regulated under strict pharmaceutical standards for medicinal purposes. These products undergo rigorous testing for purity, potency, and consistency, often requiring prescriptions and dispensed through licensed pharmacies or clinics, unlike recreational cannabis.
How is pharmaceutical marijuana different from recreational cannabis?
Pharmaceutical marijuana is precisely formulated, quality-controlled, and prescribed for specific medical conditions, often with standardized dosages and cannabinoid profiles. Recreational cannabis is typically used for non-medical purposes, has varying potencies, and is subject to less stringent quality regulations, usually sold through dispensaries or regulated retail channels.
What are the primary medical applications of pharmaceutical marijuana?
Pharmaceutical marijuana is primarily used for managing chronic pain, reducing spasticity in conditions like multiple sclerosis, controlling seizures in certain epilepsies, alleviating nausea and vomiting associated with chemotherapy, and stimulating appetite in patients with conditions like HIV/AIDS. Emerging research also explores its use in mental health and neurodegenerative disorders.
What is the growth outlook for the pharmaceutical marijuana market?
The pharmaceutical marijuana market is projected to experience substantial growth, driven by increasing global legalization for medical use, growing scientific evidence supporting its therapeutic benefits, and rising investments in research and development. This expansion is further fueled by the rising prevalence of chronic diseases and demand for alternative treatment options.
What are the main challenges facing the pharmaceutical marijuana industry?
Key challenges include inconsistent and complex regulatory frameworks across different regions, the lingering social stigma associated with cannabis, the high costs of rigorous clinical trials required for pharmaceutical approval, and difficulties in ensuring standardized quality and consistency of products. Limited long-term research data also poses a challenge to broader medical acceptance.

