Portable Spirometry Device Market
Portable Spirometry Device Market Size, Scope, Growth, Trends and By Segmentation Types, Applications, Regional Analysis and Industry Forecast (2025-2033)
Report ID : RI_700636 | Last Updated : July 26, 2025 |
Format : ![]()
![]()
![]()
![]()
Portable Spirometry Device Market Size
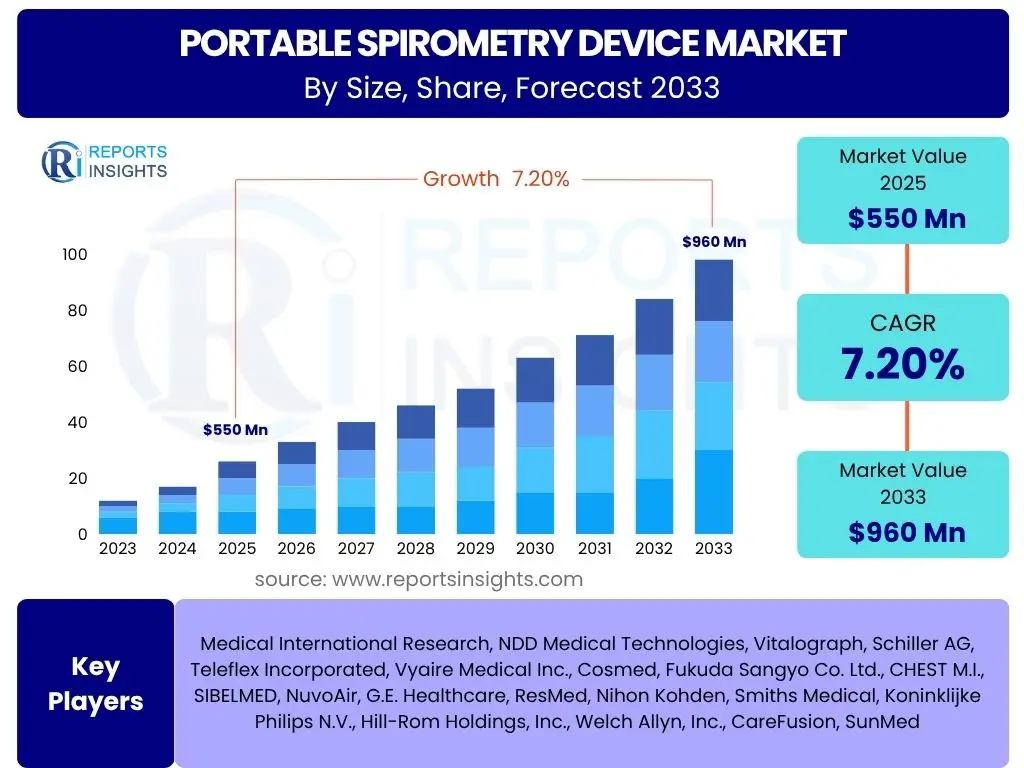
Portable Spirometry Device Market is projected to grow at a Compound annual growth rate (CAGR) of 7.2% between 2025 and 2033, valued at USD 550 million in 2025 and is projected to grow by USD 960 million by 2033 at the end of the forecast period.
Key Portable Spirometry Device Market Trends & Insights
The portable spirometry device market is undergoing significant transformation, driven by technological advancements and evolving healthcare needs. Key trends emphasize enhanced accessibility, connectivity, and integration, transforming traditional respiratory diagnostics into more dynamic and patient-centric solutions. These innovations are reshaping how respiratory conditions are managed, moving towards predictive and personalized care models.
- Miniaturization and compact design enabling greater portability.
- Integration with digital health platforms and telehealth solutions.
- Enhanced connectivity features such as Bluetooth and Wi-Fi for data transfer.
- Development of user-friendly interfaces for home use.
- Incorporation of artificial intelligence (AI) for data analysis and interpretation.
- Focus on accuracy and reproducibility of measurements in non-clinical settings.
- Rise in demand for point-of-care diagnostics for chronic respiratory diseases.
AI Impact Analysis on Portable Spirometry Device
Artificial intelligence is set to revolutionize the portable spirometry device market by enhancing diagnostic capabilities, improving user experience, and facilitating more effective disease management. AI algorithms can process complex spirometry data, identify subtle patterns indicative of respiratory conditions, and provide immediate, actionable insights, thereby reducing the burden on healthcare professionals and empowering patients with better self-management tools. The integration of AI extends beyond mere data interpretation, influencing predictive analytics, personalized therapy adjustments, and streamlined data sharing within healthcare ecosystems.
- Automated interpretation of spirometry results reducing diagnostic errors.
- Predictive analytics for early detection of respiratory exacerbations.
- Personalized feedback and coaching for patients on device usage.
- Streamlined data management and secure sharing with healthcare providers.
- Improved diagnostic accuracy and efficiency in remote settings.
- Identification of subtle trends in lung function changes over time.
- Reduced need for manual data entry and analysis.
Key Takeaways Portable Spirometry Device Market Size & Forecast
- The portable spirometry device market is poised for robust growth, driven by increasing prevalence of chronic respiratory diseases and demand for home-based care solutions.
- Technological advancements, including miniaturization, connectivity, and AI integration, are central to market expansion.
- North America and Europe currently dominate the market due to strong healthcare infrastructure and high adoption rates of advanced medical devices.
- Asia Pacific is emerging as a high-growth region, fueled by rising healthcare expenditure and increasing awareness of respiratory health.
- Opportunities exist in expanding telehealth integration, developing more cost-effective devices for emerging markets, and enhancing data security features.
- Challenges include regulatory complexities, data privacy concerns, and ensuring device accuracy in diverse user environments.
- The market's future will be characterized by a shift towards preventive care, personalized medicine, and widespread accessibility of diagnostic tools beyond traditional clinical settings.
Portable Spirometry Device Market Drivers Analysis
The portable spirometry device market is significantly propelled by several key factors that underscore its growing importance in modern healthcare. A primary driver is the escalating global burden of chronic respiratory diseases such as Chronic Obstructive Pulmonary Disease (COPD) and asthma, which necessitate continuous monitoring and early diagnosis. Concurrently, the rapidly expanding geriatric population, highly susceptible to these conditions, further fuels demand for accessible and convenient diagnostic tools. Technological advancements, particularly in device miniaturization, enhanced connectivity, and user-friendly interfaces, are making portable spirometers more appealing for both clinical and home use. The growing trend towards home healthcare and remote patient monitoring, amplified by recent global health events, has also significantly boosted the adoption of these devices, moving diagnostics out of traditional hospital settings. Furthermore, supportive government initiatives and public health campaigns aimed at early disease detection and management are creating a conducive environment for market expansion, particularly in developed economies. These collective forces are fostering a paradigm shift towards proactive respiratory health management, making portable spirometry devices indispensable tools in preventive and ongoing care.
| Drivers | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Increasing Prevalence of Chronic Respiratory Diseases: The rising global incidence of conditions like COPD, asthma, cystic fibrosis, and other lung disorders creates a continuous and growing demand for diagnostic and monitoring tools. Portable spirometers are essential for early detection and ongoing management. | +2.1% | Global, particularly North America, Europe, and Asia Pacific (China, India) due to pollution and aging populations. | Long-term (2025-2033) |
| Aging Global Population: The elderly demographic is more susceptible to respiratory ailments, necessitating frequent lung function monitoring. Portable spirometers offer a convenient solution for this vulnerable population, reducing hospital visits. | +1.8% | Europe (Germany, Italy), North America (US, Canada), and parts of Asia (Japan, South Korea). | Long-term (2025-2033) |
| Technological Advancements and Miniaturization: Continuous innovation leading to smaller, more accurate, and user-friendly devices with enhanced connectivity (Bluetooth, Wi-Fi) facilitates widespread adoption in home and point-of-care settings. | +1.5% | Developed economies (US, UK, Germany) and emerging tech hubs (South Korea, Israel). | Mid to Long-term (2025-2030) |
| Growing Demand for Home Healthcare and Telehealth: The shift from hospital-centric care to home-based monitoring and remote consultations drives the need for portable diagnostic devices, making healthcare more accessible and cost-effective. | +1.3% | North America, Europe, and increasingly in Asia Pacific (Australia, Singapore). | Mid-term (2025-2028) |
| Favorable Government Initiatives and Reimbursement Policies: Increased government funding for respiratory health programs, awareness campaigns, and supportive reimbursement policies for home diagnostic tests encourage adoption and market growth. | +0.5% | North America (US), Europe (UK, Germany), and select Asian countries (Japan). | Short to Mid-term (2025-2027) |
Portable Spirometry Device Market Restraints Analysis
Despite the promising growth trajectory, the portable spirometry device market faces several significant restraints that could impede its full potential. One major hurdle is the relatively high cost associated with advanced portable spirometers, which can be prohibitive for individuals in low-income regions or for healthcare systems with constrained budgets. This financial barrier often limits widespread adoption, particularly in developing countries where healthcare infrastructure is less robust. Furthermore, concerns regarding the accuracy and calibration stability of portable devices, especially when compared to gold-standard laboratory spirometers, can create skepticism among healthcare professionals and lead to underutilization. The complexity of operating and interpreting results from some portable devices without professional guidance can also be a deterrent for home users, leading to improper usage and unreliable data. Additionally, stringent regulatory approval processes in various regions can delay market entry for new innovations, while a lack of comprehensive reimbursement policies in some healthcare systems may reduce the economic incentive for both providers and patients to adopt these devices. Addressing these restraints through cost-effective innovations, improved user interfaces, and supportive regulatory frameworks will be crucial for sustained market expansion.
| Restraints | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| High Cost of Advanced Portable Spirometers: The initial investment for high-precision portable spirometers can be substantial, limiting affordability for individual patients and healthcare facilities with budget constraints, especially in developing regions. | -0.9% | Developing nations (Africa, Southeast Asia), and price-sensitive markets globally. | Long-term (2025-2033) |
| Accuracy and Calibration Concerns: Despite technological advancements, some portable devices may face challenges in maintaining consistent accuracy and requiring frequent calibration, potentially leading to clinician skepticism and affecting diagnostic reliability. | -0.7% | Global, especially in clinical settings requiring high precision. | Mid-term (2025-2029) |
| Lack of Awareness and Training in Emerging Markets: Insufficient public and professional awareness about the benefits of portable spirometry, coupled with a lack of adequate training for device usage and data interpretation, hampers adoption in less developed regions. | -0.5% | Africa, Latin America, and parts of Asia Pacific. | Long-term (2025-2033) |
| Stringent Regulatory Approval Processes: Obtaining necessary approvals from regulatory bodies such as the FDA and EMA can be time-consuming and expensive, delaying the market entry of innovative devices and increasing R&D costs for manufacturers. | -0.3% | North America, Europe, and other highly regulated markets. | Short to Mid-term (2025-2027) |
Portable Spirometry Device Market Opportunities Analysis
The portable spirometry device market is rich with opportunities that promise significant future expansion and innovation. A substantial opportunity lies in the further integration of these devices with smart platforms and mobile applications, creating comprehensive digital health ecosystems that enhance patient engagement and streamline data sharing with healthcare providers. The untapped potential in emerging economies, characterized by improving healthcare infrastructure, rising disposable incomes, and increasing awareness of respiratory diseases, represents a lucrative growth avenue for manufacturers. Research and development into novel sensor technologies and advanced algorithms, particularly those leveraging artificial intelligence and machine learning, can lead to more accurate, sensitive, and user-friendly devices with enhanced diagnostic capabilities. Furthermore, the burgeoning field of personalized medicine offers an opportunity to tailor spirometry monitoring to individual patient needs, optimizing treatment plans and improving patient outcomes. Strategic partnerships between device manufacturers, telehealth providers, and pharmaceutical companies can also unlock new distribution channels and integrated care models. By capitalizing on these opportunities, market players can drive innovation, expand market reach, and establish new standards in respiratory care, transforming how lung health is monitored and managed globally.
| Opportunities | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Integration with Smart Devices and Mobile Applications: Developing portable spirometers that seamlessly connect with smartphones and tablets can enhance user experience, provide real-time data analysis, and facilitate remote monitoring and data sharing. | +2.0% | Global, particularly in regions with high smartphone penetration (Asia Pacific, North America, Europe). | Short to Mid-term (2025-2028) |
| Expansion into Emerging Markets: Untapped potential in countries with large populations, growing healthcare expenditure, and increasing awareness of respiratory diseases (e.g., China, India, Brazil) offers significant market penetration opportunities. | +1.8% | Asia Pacific, Latin America, and Middle East & Africa. | Long-term (2027-2033) |
| Advanced Sensor Technologies and AI/ML Integration: Investment in R&D for novel sensors, artificial intelligence, and machine learning algorithms can lead to more accurate, predictive, and user-friendly devices, enhancing diagnostic capabilities and personalized care. | +1.5% | Developed economies with strong R&D infrastructure (US, Germany, Japan). | Mid to Long-term (2026-2033) |
| Focus on Personalized Medicine and Preventive Care: Tailoring spirometry monitoring solutions to individual patient profiles and integrating them into preventive health strategies can drive adoption and improve long-term patient outcomes. | +1.0% | North America and Europe, with increasing relevance globally. | Mid-term (2025-2030) |
Portable Spirometry Device Market Challenges Impact Analysis
The portable spirometry device market faces distinct challenges that require strategic navigation for sustained growth and broader adoption. One significant hurdle is ensuring data security and patient privacy, as these devices often transmit sensitive health information through connected platforms. Robust cybersecurity measures are essential to build trust and comply with stringent data protection regulations. Another challenge stems from the inherent need for consistent device calibration and ongoing maintenance to guarantee accurate and reliable results, particularly for devices used in home settings without professional oversight. Competition from traditional, more established spirometry systems, alongside the emergence of alternative diagnostic methods for respiratory conditions, presents a competitive pressure that mandates continuous innovation and differentiation. Furthermore, variability in reimbursement policies across different healthcare systems and regions can impact the commercial viability of portable spirometry devices, influencing both physician prescribing patterns and patient access. Educating both healthcare professionals and end-users on proper device usage, maintenance, and the interpretation of results also remains a crucial challenge, especially in diverse demographic and technological landscapes. Overcoming these challenges will necessitate a multi-faceted approach involving technological advancements, regulatory compliance, educational initiatives, and strategic market positioning.
| Challenges | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Data Security and Patient Privacy Concerns: The transmission and storage of sensitive patient spirometry data via connected devices raise significant concerns about cybersecurity breaches and compliance with strict data privacy regulations (e.g., HIPAA, GDPR). | -0.8% | Global, particularly in highly regulated regions like North America and Europe. | Long-term (2025-2033) |
| Calibration and Maintenance Issues for Home Users: Ensuring the regular and accurate calibration of portable devices, along with proper maintenance by non-medical professionals in home settings, is challenging and can impact data reliability. | -0.6% | Global, especially for devices intended for widespread home use. | Mid-term (2025-2030) |
| Competition from Alternative Diagnostic Methods: The market faces competition from other diagnostic tools for respiratory conditions or from traditional, less portable but highly accurate spirometry systems in clinical settings, requiring differentiation. | -0.5% | Global, especially in developed healthcare markets. | Short to Mid-term (2025-2028) |
| Variability in Reimbursement Policies: Inconsistent or inadequate reimbursement coverage for portable spirometry tests across different regions and healthcare systems can limit physician prescription and patient access, affecting market growth. | -0.3% | North America (US), Europe, and varying widely by country. | Long-term (2025-2033) |
Portable Spirometry Device Market - Updated Report Scope
This comprehensive market research report provides an in-depth analysis of the Portable Spirometry Device Market, offering critical insights into its current landscape, historical performance, and future growth projections. The report is meticulously designed to support strategic decision-making for stakeholders across the healthcare industry, including manufacturers, suppliers, healthcare providers, and investors. It covers key market dynamics, including detailed assessments of market size and forecast, significant trends, the impact of artificial intelligence, and a thorough analysis of drivers, restraints, opportunities, and challenges. Furthermore, the report presents a granular segmentation of the market and highlights regional performance, concluding with profiles of key industry players to provide a holistic view of the competitive landscape. This updated scope ensures that the analysis remains relevant and forward-looking, addressing the evolving needs of the respiratory diagnostics sector and reflecting the latest market intelligence.
| Report Attributes | Report Details |
|---|---|
| Base Year | 2024 |
| Historical Year | 2019 to 2023 |
| Forecast Year | 2025 - 2033 |
| Market Size in 2025 | USD 550 Million |
| Market Forecast in 2033 | USD 960 Million |
| Growth Rate | 7.2% CAGR from 2025 to 2033 |
| Number of Pages | 265 |
| Key Trends |
|
| Segments Covered |
|
| Key Companies Covered | Medical International Research, NDD Medical Technologies, Vitalograph, Schiller AG, Teleflex Incorporated, Vyaire Medical Inc., Cosmed, Fukuda Sangyo Co. Ltd., CHEST M.I., SIBELMED, NuvoAir, G.E. Healthcare, ResMed, Nihon Kohden, Smiths Medical, Koninklijke Philips N.V., Hill-Rom Holdings, Inc., Welch Allyn, Inc., CareFusion, SunMed |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Speak to Analyst | Avail customised purchase options to meet your exact research needs. Request For Analyst Or Customization |
Segmentation Analysis
The Portable Spirometry Device Market is comprehensively segmented to provide a detailed understanding of its diverse components and drivers. This segmentation allows for targeted analysis of market dynamics across various dimensions, including device type, underlying technology, clinical application, and end-user adoption patterns. Such granular insights are crucial for identifying niche markets, understanding specific demand drivers, and formulating precise strategic approaches. Each segment reflects unique characteristics and growth potentials, contributing to the overall market landscape and influencing investment decisions.
- By Type: This segment differentiates portable spirometry devices based on their form factor and typical use environments.
- Handheld Portable Spirometers: Compact, lightweight devices designed for maximum portability and ease of use in home settings, point-of-care, or remote monitoring scenarios. They prioritize convenience and simplicity.
- Tabletop/Desktop Portable Spirometers: While still portable, these devices are generally larger than handheld units, offering more advanced features, higher accuracy, and often a larger display, suitable for small clinics, research settings, or more comprehensive home use.
- By Technology: This segment categorizes devices based on the fundamental mechanism used to measure lung function.
- Volume-Based Spirometers: Measure lung volumes directly by assessing the displacement of a piston or bellows, known for their accuracy and often used as a gold standard in clinical settings.
- Flow-Based Spirometers: Measure the rate of airflow during breathing and then integrate this flow over time to derive lung volumes. They are commonly used in portable devices due to their compact design and ease of use.
- Turbine-Based Spirometers: A subset of flow-based spirometers that use a turbine or rotating vane to measure airflow. They are known for their robustness and are widely adopted in both portable and clinical settings.
- By Application: This segment focuses on the specific medical conditions or clinical needs for which portable spirometry devices are primarily used.
- Chronic Obstructive Pulmonary Disease (COPD): A major application area due to the chronic nature of the disease, requiring regular monitoring of lung function to manage exacerbations and disease progression.
- Asthma: Essential for diagnosing asthma, monitoring its severity, and assessing the effectiveness of treatment regimens, especially in pediatric and adult patients.
- Cystic Fibrosis: Portable spirometers are vital for monitoring lung function decline in patients with cystic fibrosis, enabling timely interventions.
- Pulmonary Fibrosis: Used to track the progression of restrictive lung diseases like pulmonary fibrosis, providing critical data for disease management.
- Other Respiratory Conditions: Includes applications for various less common respiratory ailments, post-operative lung function assessment, and general respiratory health screening.
- By End-User: This segment differentiates the market based on the primary settings or entities utilizing portable spirometry devices.
- Hospitals: Used for initial diagnostics, pre- and post-operative assessments, and monitoring of inpatients with respiratory issues, often complementing larger clinical spirometers.
- Clinics: Widely adopted in general practitioner clinics, specialized pulmonology clinics, and urgent care centers for routine lung function assessments.
- Diagnostic Centers: Facilities specifically dedicated to diagnostic testing utilize portable spirometers for a range of respiratory function tests.
- Home Care Settings: A rapidly growing segment, driven by the increasing need for remote patient monitoring, chronic disease management, and patient convenience.
- Research & Academic Institutions: Employed in clinical trials, medical research, and academic studies related to respiratory physiology and disease.
Regional Highlights
The global portable spirometry device market exhibits distinct growth patterns across various geographical regions, influenced by healthcare infrastructure, disease prevalence, technological adoption, and regulatory landscapes. Understanding these regional dynamics is crucial for market participants to identify lucrative opportunities and tailor their strategies effectively.
- North America: This region consistently holds a dominant share in the portable spirometry device market. The United States and Canada are key contributors, driven by a high prevalence of chronic respiratory diseases, advanced healthcare infrastructure, significant awareness regarding early diagnosis, and robust adoption of telehealth and home healthcare solutions. High healthcare expenditure, favorable reimbursement policies, and the presence of key market players also bolster market growth here.
- Europe: Europe represents another significant market for portable spirometry devices, propelled by an aging population highly susceptible to respiratory ailments, well-established healthcare systems, and increasing patient awareness. Countries like Germany, the United Kingdom, France, and Italy are leading the market due to strong government support for chronic disease management programs and the growing integration of digital health technologies in clinical practice.
- Asia Pacific (APAC): The Asia Pacific region is projected to be the fastest-growing market for portable spirometry devices. This rapid expansion is attributed to the large patient pool with respiratory conditions, particularly in populous countries like China and India, increasing healthcare expenditure, improving healthcare infrastructure, and rising disposable incomes. The growing awareness about lung health, coupled with governmental initiatives to combat air pollution and promote early disease detection, further fuels market growth in this region. Japan, South Korea, and Australia also demonstrate strong potential due driven by technological adoption and growing medical tourism.
- Latin America: This region is experiencing steady growth in the portable spirometry device market, primarily due to increasing awareness of respiratory diseases, improving access to healthcare services, and a rising prevalence of risk factors like smoking and environmental pollution. Countries such as Brazil, Mexico, and Argentina are key markets, driven by government efforts to enhance public health and the gradual adoption of portable diagnostic solutions.
- Middle East and Africa (MEA): The MEA market for portable spirometry devices is gradually expanding. Growth is primarily driven by rising healthcare investments, an increasing burden of non-communicable diseases including respiratory conditions, and efforts to modernize healthcare facilities. However, market growth in some parts of the region is constrained by limited healthcare infrastructure and lower per capita healthcare spending. South Africa, Saudi Arabia, and UAE are notable markets due to higher healthcare spending and medical tourism.
Top Key Players:
The market research report covers the analysis of key stake holders of the Portable Spirometry Device Market. Some of the leading players profiled in the report include -
- Medical International Research
- NDD Medical Technologies
- Vitalograph
- Schiller AG
- Teleflex Incorporated
- Vyaire Medical Inc.
- Cosmed
- Fukuda Sangyo Co. Ltd.
- CHEST M.I.
- SIBELMED
- NuvoAir
- G.E. Healthcare
- ResMed
- Nihon Kohden
- Smiths Medical
- Koninklijke Philips N.V.
- Hill-Rom Holdings, Inc.
- Welch Allyn, Inc.
- CareFusion
- SunMed
Frequently Asked Questions:
What is a portable spirometry device?
A portable spirometry device is a compact, handheld or tabletop instrument designed to measure lung function by assessing the volume and flow of air a person can inhale and exhale. These devices are crucial for diagnosing, monitoring, and managing various respiratory conditions such as asthma, COPD, and cystic fibrosis, often providing real-time results outside of traditional clinical settings.
Why are portable spirometry devices important for respiratory health?
Portable spirometry devices are vital for respiratory health because they enable convenient and frequent monitoring of lung function, facilitating early detection of respiratory diseases, tracking disease progression, and evaluating treatment effectiveness. Their portability allows patients to perform tests at home, improving adherence to monitoring protocols and providing healthcare professionals with continuous, actionable data for personalized care.
What are the key benefits of using portable spirometry devices?
Key benefits of portable spirometry devices include enhanced accessibility to lung function testing, enabling remote patient monitoring and telehealth consultations. They offer convenience for patients, reduce the need for frequent hospital visits, and empower individuals to actively participate in managing their respiratory health. These devices also provide immediate results, aiding in quicker diagnostic decisions and timely therapeutic adjustments.
How is AI impacting the portable spirometry device market?
Artificial intelligence is significantly impacting the portable spirometry device market by enhancing data interpretation, providing automated diagnostic support, and enabling predictive analytics for respiratory conditions. AI algorithms can analyze complex spirometry waveforms, identify subtle anomalies, and offer personalized feedback, thereby improving diagnostic accuracy, streamlining workflows for healthcare providers, and facilitating proactive disease management for patients.
What is the market forecast for portable spirometry devices?
The portable spirometry device market is projected to grow at a Compound Annual Growth Rate (CAGR) of 7.2% between 2025 and 2033. Valued at USD 550 million in 2025, the market is expected to reach approximately USD 960 million by the end of the forecast period in 2033, driven by increasing prevalence of respiratory diseases, technological advancements, and the growing adoption of home healthcare solutions.