Minimally Invasive Medical Device Market
Minimally Invasive Medical Device Market Size, Scope, Growth, Trends and By Segmentation Types, Applications, Regional Analysis and Industry Forecast (2025-2033)
Report ID : RI_700229 | Last Updated : July 23, 2025 |
Format : ![]()
![]()
![]()
![]()
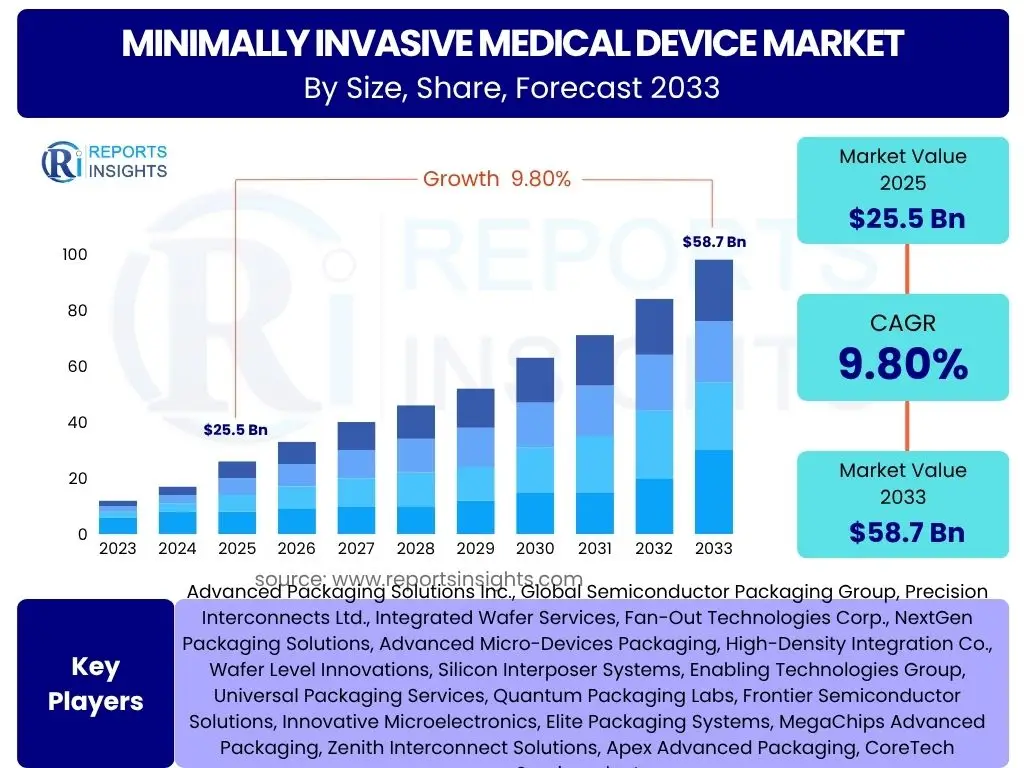
Minimally Invasive Medical Device Market is projected to grow at a Compound annual growth rate (CAGR) of 9.8% between 2025 and 2033, current valued at USD 25.5 billion in 2025 and is projected to grow by USD 58.7 billion by 2033 the end of the forecast period.
Key Minimally Invasive Medical Device Market Trends & Insights
The Minimally Invasive Medical Device (MIMD) market is currently experiencing a transformative phase, driven by continuous innovation and a global shift towards less invasive surgical interventions. Key trends indicate a strong preference among patients and healthcare providers for procedures that offer reduced pain, faster recovery times, and lower complication rates compared to traditional open surgeries. This preference is fueling the adoption of advanced robotic systems, sophisticated imaging techniques, and highly specialized instrumentation, redefining surgical standards worldwide.
Furthermore, the market is profoundly influenced by an aging global population, which presents an increasing incidence of chronic diseases requiring surgical management. Technological convergence, particularly the integration of artificial intelligence and machine learning, is creating new possibilities for enhanced precision, improved diagnostic capabilities, and personalized treatment approaches. This convergence, combined with expanding healthcare infrastructure in emerging economies, points towards a robust and dynamic growth trajectory for the MIMD sector.
The leading trends shaping the Minimally Invasive Medical Device market include:
- Increasing adoption of robotic-assisted surgery systems.
- Advancements in visualization technologies, including 3D and 4K imaging.
- Growth in single-port and natural orifice transluminal endoscopic surgery (NOTES).
- Development of smaller, more flexible, and multi-functional instruments.
- Rising demand for disposable minimally invasive devices for infection control.
- Integration of smart sensors and real-time feedback mechanisms into devices.
- Expansion of minimally invasive procedures across various therapeutic areas.
- Growing focus on personalized medicine and patient-specific surgical planning.
AI Impact Analysis on Minimally Invasive Medical Device
The integration of Artificial Intelligence (AI) is rapidly revolutionizing the Minimally Invasive Medical Device (MIMD) landscape, offering unprecedented opportunities for enhanced precision, improved patient outcomes, and operational efficiency. AI algorithms are transforming every stage of minimally invasive procedures, from pre-operative planning and intra-operative guidance to post-operative recovery monitoring. This technological synergy allows for more informed decision-making by surgeons, reduced human error, and the potential for greater standardization in complex procedures, ultimately leading to superior healthcare delivery.
AI's influence extends to surgical robotics, where it powers autonomous navigation, enhances tremor reduction, and provides real-time analytics to guide surgical maneuvers. In diagnostic imaging, AI-driven solutions are improving the accuracy and speed of pathology detection, enabling earlier and more precise interventions. Furthermore, AI is crucial in predictive analytics for patient risk assessment and optimizing post-operative care pathways, signaling a paradigm shift towards intelligent and adaptive minimally invasive solutions. The following bullet points highlight key areas of AI's transformative impact:
- Enhanced precision and stability in robotic-assisted surgeries.
- Real-time image analysis and augmented reality overlay for improved visualization.
- Predictive analytics for patient selection and risk stratification in MIMD.
- Automated anomaly detection in endoscopic and laparoscopic imaging.
- Intelligent instrument design and optimization through machine learning.
- Training and simulation platforms leveraging AI for surgeon skill development.
- Streamlined workflow and reduced procedural times through AI-driven automation.
- Personalized surgical planning based on AI analysis of patient-specific data.
Key Takeaways Minimally Invasive Medical Device Market Size & Forecast
- The Minimally Invasive Medical Device market is projected for substantial growth between 2025 and 2033.
- Anticipated to achieve a Compound Annual Growth Rate (CAGR) of 9.8% over the forecast period.
- Market size estimated at USD 25.5 billion in 2025.
- Projected to reach an impressive USD 58.7 billion by the end of 2033.
- Significant expansion driven by technological innovation and increasing procedural adoption.
- The forecast reflects a sustained demand for less invasive surgical options globally.
- Market growth is consistently strong across various product segments and therapeutic applications.
Minimally Invasive Medical Device Market Drivers Analysis
The Minimally Invasive Medical Device (MIMD) market is propelled by a confluence of powerful drivers that collectively contribute to its robust expansion. A primary catalyst is the accelerating global demographic shift, characterized by an aging population. As individuals live longer, the prevalence of age-related chronic diseases, such as cardiovascular conditions, orthopedic ailments, and various cancers, significantly increases. These conditions often necessitate surgical interventions, and the inherent benefits of minimally invasive procedures—reduced trauma, quicker recovery, and fewer post-operative complications—make them the preferred choice for both patients and healthcare providers.
Another significant driver is the continuous advancement in medical technology. Innovations in robotic surgery, advanced imaging techniques, endoscopic instruments, and specialized energy devices are constantly expanding the scope and safety of minimally invasive procedures. These technological leaps enable surgeons to perform complex operations with unprecedented precision and control, making MIMD applicable to a wider range of therapeutic areas. Furthermore, growing patient awareness and preference for less invasive options, coupled with supportive reimbursement policies in developed economies, are further stimulating market demand.
| Drivers | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Rising Global Geriatric Population & Chronic Disease Incidence | +2.5% | North America, Europe, Asia Pacific (Japan, China) | Long-term (2025-2033) |
| Technological Advancements in Surgical Robotics & Imaging | +2.2% | Global, particularly North America, Europe | Continuous (2025-2033) |
| Increasing Patient Preference for Less Invasive Procedures | +1.8% | Developed & Emerging Economies | Mid to Long-term (2025-2033) |
| Favorable Reimbursement Policies & Healthcare Reforms | +1.5% | North America, Western Europe | Mid-term (2025-2030) |
| Expansion of Healthcare Infrastructure in Emerging Markets | +1.0% | Asia Pacific (India, China), Latin America, MEA | Long-term (2027-2033) |
Minimally Invasive Medical Device Market Restraints Analysis
Despite its significant growth trajectory, the Minimally Invasive Medical Device (MIMD) market faces several notable restraints that could temper its expansion. One of the most prominent challenges is the high cost associated with MIMD procedures and equipment. Advanced robotic systems, specialized instruments, and cutting-edge imaging technologies require substantial initial investments from healthcare facilities. This high capital expenditure, coupled with the ongoing costs of maintenance and training, can be prohibitive for many hospitals, especially in regions with limited healthcare budgets or less developed infrastructure, thereby restricting broader adoption.
Furthermore, the market's growth is constrained by the stringent regulatory landscape governing medical devices. Obtaining approvals from regulatory bodies such as the FDA in the United States or the EMA in Europe is a lengthy, complex, and expensive process, involving rigorous testing and clinical trials. These extensive regulatory hurdles can delay product launches, stifle innovation, and increase the overall cost of bringing new devices to market. Additionally, the steep learning curve and specialized training required for surgeons and medical staff to competently perform minimally invasive procedures also act as a significant restraint, limiting the pool of qualified practitioners and slowing the widespread implementation of these techniques.
| Restraints | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| High Cost of Minimally Invasive Devices & Procedures | -1.8% | Global, particularly developing economies | Long-term (2025-2033) |
| Stringent Regulatory Approval Processes | -1.5% | North America, Europe | Continuous (2025-2033) |
| Lack of Skilled Professionals & Extensive Training Requirements | -1.2% | Global, more pronounced in emerging markets | Mid to Long-term (2025-2033) |
| Potential for Product Recalls & Safety Concerns | -0.8% | Global | Intermittent (as incidents occur) |
Minimally Invasive Medical Device Market Opportunities Analysis
The Minimally Invasive Medical Device (MIMD) market is poised for significant future growth, driven by an array of emerging opportunities that promise to reshape its landscape. A key area of potential lies in the vast untapped markets within emerging economies. As healthcare infrastructure improves and disposable incomes rise in regions like Asia Pacific, Latin America, and the Middle East & Africa, there is a burgeoning demand for advanced medical technologies. Local governments and private healthcare providers are increasingly investing in modern facilities and adopting advanced surgical techniques, creating fertile ground for MIMD market expansion beyond traditional developed markets.
Another substantial opportunity stems from the continuous innovation in technological convergence, particularly the integration of Artificial Intelligence (AI), augmented reality (AR), and virtual reality (VR) with minimally invasive procedures. These technologies hold the promise of enhancing surgical precision, offering immersive training environments, and enabling real-time diagnostic capabilities, thereby broadening the application scope and improving outcomes. Furthermore, the increasing shift towards personalized medicine and outpatient surgical centers presents avenues for specialized MIMD solutions, catering to specific patient needs and enabling cost-effective, patient-centric care models.
| Opportunities | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Expansion into Emerging Markets & Underserved Regions | +2.0% | Asia Pacific, Latin America, MEA | Long-term (2025-2033) |
| Integration of AI, AR, VR, and Robotics in MIMD | +1.7% | Global, R&D intensive regions (North America, Europe) | Continuous Innovation (2025-2033) |
| Growth of Ambulatory Surgical Centers (ASCs) & Outpatient Procedures | +1.5% | North America, Western Europe | Mid-term (2025-2030) |
| Development of Next-Generation Biocompatible Materials | +1.0% | Global, R&D hubs | Long-term (2027-2033) |
Minimally Invasive Medical Device Market Challenges Impact Analysis
Despite its promising outlook, the Minimally Invasive Medical Device (MIMD) market is confronted by several complex challenges that demand strategic attention from industry stakeholders. One significant hurdle is the escalating intensity of competition among market players. With numerous established companies and emerging startups vying for market share, there is immense pressure on pricing, innovation cycles, and differentiation. This intense rivalry can lead to thinner profit margins, increased marketing expenditures, and a constant need for research and development to maintain a competitive edge, posing a threat to smaller or less innovative firms.
Another critical challenge is the inherent complexity associated with the manufacturing and supply chain of advanced medical devices. These devices often require highly specialized components, precision engineering, and adherence to stringent quality controls. Disruptions in global supply chains, such as those experienced during recent global health crises, can severely impact production, lead to shortages, and escalate costs. Furthermore, ensuring data security and addressing cybersecurity threats related to interconnected medical devices and patient information present a growing concern, requiring robust protective measures and continuous vigilance to prevent breaches and maintain patient trust.
| Challenges | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Intense Competition and Pricing Pressures | -1.5% | Global, particularly North America, Europe | Continuous (2025-2033) |
| Supply Chain Vulnerabilities & Raw Material Scarcity | -1.2% | Global | Short to Mid-term (2025-2028) |
| Cybersecurity Threats & Data Privacy Concerns | -1.0% | Global | Continuous (2025-2033) |
| Ethical Considerations & Public Acceptance of Advanced Technologies | -0.7% | Developed Economies | Long-term (2027-2033) |
Minimally Invasive Medical Device Market - Updated Report Scope
This comprehensive market research report offers an in-depth analysis of the Minimally Invasive Medical Device market, providing critical insights into its current dynamics, historical performance, and future growth trajectories. It encompasses a detailed examination of market size, trends, drivers, restraints, opportunities, and challenges across various segments and key geographical regions. The report is designed to equip stakeholders with actionable intelligence for strategic decision-making, offering a granular view of market evolution from 2019 to 2033.
| Report Attributes | Report Details |
|---|---|
| Base Year | 2024 |
| Historical Year | 2019 to 2023 |
| Forecast Year | 2025 - 2033 |
| Market Size in 2025 | USD 25.5 Billion |
| Market Forecast in 2033 | USD 58.7 Billion |
| Growth Rate | 9.8% CAGR from 2025 to 2033 |
| Number of Pages | 267 |
| Key Trends |
|
| Segments Covered |
|
| Key Companies Covered | Medtronic, Johnson & Johnson, Stryker, Abbott, Boston Scientific, Zimmer Biomet, B. Braun Melsungen, Smith & Nephew, ConMed Corporation, Karl Storz, Olympus Corporation, Intuitive Surgical, Edwards Lifesciences, Teleflex Incorporated, Getinge AB, Asahi Kasei Corporation, Ethicon, Hologic Inc., AngioDynamics, Baxter International |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Speak to Analyst | Avail customised purchase options to meet your exact research needs. Request For Analyst Or Customization |
Segmentation Analysis
:The Minimally Invasive Medical Device market is comprehensively segmented to provide a granular understanding of its diverse components and sub-sectors. This segmentation allows for a detailed analysis of market dynamics, growth drivers, and opportunities across various product types, application areas, and end-user categories. Understanding these segments is crucial for stakeholders to identify lucrative niches, develop targeted strategies, and forecast market performance accurately.
The segmentation reflects the multifaceted nature of the MIMD market, encompassing a wide array of devices used for diagnosis, treatment, and monitoring across numerous medical specialties. From intricate surgical instruments designed for precision to advanced monitoring devices that provide real-time patient data, each segment plays a vital role in the overall market landscape. Furthermore, the categorization by end-user highlights the primary settings where these devices are utilized, offering insights into demand patterns from hospitals, ambulatory surgical centers, and specialized clinics.
- By Product: This segment includes the various types of devices utilized in minimally invasive procedures.
- Surgical Instruments: Comprising tools like forceps, scissors, retractors, trocars, and specialized suturing devices essential for precise surgical maneuvers.
- Monitoring Devices: Includes equipment such as cardiac monitors, vital sign monitors, and other physiological monitoring systems critical during and after procedures.
- Ablation Devices: Devices used for tissue destruction through heat, cold, or other energy forms, including radiofrequency ablation, cryoablation, and microwave ablation.
- Endoscopic Devices: Encompasses various types of endoscopes (e.g., gastroscopes, colonoscopes, bronchoscopes), laparoscopes, arthroscopes, and related accessories like insufflators and light sources.
- Electrosurgical Devices: Tools that use high-frequency electrical current for cutting, coagulating, and dissecting tissue, such as electrosurgical generators and electrodes.
- Others: Includes a range of miscellaneous devices like access devices, guidewires, catheters, and fixation devices that support minimally invasive surgeries.
- By Application: This segment categorizes the market based on the medical specialties and conditions where minimally invasive devices are applied.
- Cardiology: Procedures related to heart and vascular conditions, including angioplasty, stent placement, and cardiac ablation.
- Orthopedics: Surgical interventions for musculoskeletal system conditions, such as arthroscopy, joint repair, and spinal procedures.
- Urology: Procedures addressing urinary tract and reproductive system disorders, including prostatectomy and nephrectomy.
- General Surgery: A broad category encompassing various abdominal, gastrointestinal, and other common surgical procedures.
- Gynecology: Procedures related to female reproductive health, such as hysterectomy and fibroid removal.
- Neurology: Surgical interventions for brain, spinal cord, and nervous system conditions, including neuroendoscopy.
- Pulmonology: Procedures involving the respiratory system, such as bronchoscopy and thoracoscopy.
- Others: Includes applications in ENT (ear, nose, and throat), ophthalmology, and other emerging areas.
- By End-User: This segment identifies the primary healthcare settings where minimally invasive medical devices are extensively used.
- Hospitals: The largest end-user segment, equipped with advanced infrastructure and specialists for a wide range of complex procedures.
- Ambulatory Surgical Centers (ASCs): Facilities offering outpatient surgical services, increasingly adopting MIMD for their cost-effectiveness and patient convenience.
- Specialty Clinics: Clinics focusing on specific medical disciplines (e.g., orthopedic clinics, urology clinics) that perform specialized minimally invasive procedures.
- Others: Includes research institutes, diagnostic centers, and academic medical centers involved in the development and application of MIMD.
Regional Highlights
Regional analysis is a critical component of understanding the global Minimally Invasive Medical Device (MIMD) market, as geographical disparities significantly influence market growth, adoption rates, and technological advancements. Each region presents unique market dynamics, shaped by factors such as healthcare infrastructure development, demographic trends, regulatory frameworks, and economic conditions. A deep dive into these regional nuances reveals where current demand is concentrated and where future opportunities are most likely to emerge, offering strategic insights for market participants.
North America, particularly the United States, stands out as a leading region due to its highly advanced healthcare system, robust investment in research and development, and early adoption of innovative medical technologies. Europe also holds a significant share, driven by an aging population, established healthcare systems, and increasing patient awareness. However, the Asia Pacific region is rapidly emerging as the fastest-growing market, propelled by expanding healthcare access, rising disposable incomes, and a growing medical tourism industry. Latin America, the Middle East, and Africa are also showing promising growth, albeit from a lower base, as their healthcare systems evolve and embrace modern surgical techniques.
- North America: This region dominates the Minimally Invasive Medical Device market, primarily due to:
- High prevalence of chronic diseases and an aging population.
- Robust healthcare infrastructure and significant healthcare expenditure.
- Early adoption of advanced technologies, including robotic surgery and advanced imaging.
- Presence of key market players and strong R&D activities.
- Favorable reimbursement policies and government support for innovative treatments.
- Europe: A significant market for MIMD, characterized by:
- Well-established healthcare systems and high patient awareness regarding minimally invasive procedures.
- Aging demographics contributing to a higher incidence of surgical conditions.
- Stringent regulatory standards ensuring high-quality device development and adoption.
- Increasing investments in digital health and surgical automation.
- Focus on reducing hospital stays and improving patient outcomes.
- Asia Pacific (APAC): Expected to exhibit the highest growth rate during the forecast period due to:
- Rapidly developing healthcare infrastructure and increasing healthcare spending.
- Large patient pool and growing medical tourism in countries like India and China.
- Rising prevalence of lifestyle-related diseases requiring surgical intervention.
- Increasing awareness and affordability of advanced medical treatments.
- Government initiatives promoting domestic manufacturing and healthcare access.
- Latin America: Demonstrates steady growth influenced by:
- Improving economic conditions and expanding healthcare access in key countries like Brazil and Mexico.
- Growing demand for advanced medical treatments as healthcare systems modernize.
- Increasing foreign direct investment in the healthcare sector.
- Development of local expertise and training in minimally invasive techniques.
- Middle East and Africa (MEA): Emerging as a market with considerable potential, driven by:
- Increasing government investments in healthcare infrastructure and medical tourism.
- Rising prevalence of chronic diseases and a growing expatriate population.
- Adoption of Western healthcare standards and advanced medical technologies in oil-rich nations.
- Strategic collaborations and partnerships with international medical device manufacturers.
Top Key Players:
The market research report covers the analysis of key stake holders of the Minimally Invasive Medical Device Market. Some of the leading players profiled in the report include -:- Medtronic
- Johnson & Johnson
- Stryker
- Abbott
- Boston Scientific
- Zimmer Biomet
- B. Braun Melsungen
- Smith & Nephew
- ConMed Corporation
- Karl Storz
- Olympus Corporation
- Intuitive Surgical
- Edwards Lifesciences
- Teleflex Incorporated
- Getinge AB
- Asahi Kasei Corporation
- Ethicon
- Hologic Inc.
- AngioDynamics
- Baxter International
Frequently Asked Questions:
What is a Minimally Invasive Medical Device?
A Minimally Invasive Medical Device (MIMD) refers to instruments and equipment designed to perform surgical procedures through small incisions or natural body openings, minimizing trauma to the body. This approach leads to less pain, reduced scarring, shorter hospital stays, and faster recovery times for patients compared to traditional open surgery. Examples include endoscopes, laparoscopes, robotic surgical systems, and specialized instruments like trocars and energy devices.
Why is the Minimally Invasive Medical Device market experiencing significant growth?
The MIMD market is growing rapidly due to several key factors: an aging global population with increasing prevalence of chronic diseases requiring surgery, continuous technological advancements (e.g., robotics, advanced imaging), rising patient preference for less painful and faster-recovering procedures, and supportive reimbursement policies in many developed healthcare systems. These factors collectively drive higher adoption rates and expanded application of MIMD technologies.
What are the key technological trends influencing the MIMD market?
Key technological trends include the proliferation of robotic-assisted surgery systems, advancements in 3D and 4K visualization technologies for enhanced surgical views, the integration of Artificial Intelligence (AI) and Machine Learning (ML) for surgical planning and real-time guidance, and the development of smaller, more flexible, and multi-functional instruments. Additionally, single-port surgery and the use of natural orifice transluminal endoscopic surgery (NOTES) are emerging trends enhancing procedural innovation.
What are the primary challenges faced by the Minimally Invasive Medical Device market?
The MIMD market faces several challenges, including the high cost associated with advanced devices and procedures, which can limit adoption, particularly in developing regions. Stringent and time-consuming regulatory approval processes also pose a significant hurdle. Furthermore, a shortage of skilled professionals trained in minimally invasive techniques and potential cybersecurity threats to interconnected medical devices present ongoing challenges for market expansion and patient safety.
Which regions are key contributors to the Minimally Invasive Medical Device market?
North America currently leads the Minimally Invasive Medical Device market due to its advanced healthcare infrastructure and high adoption of innovative technologies. Europe also holds a substantial market share, driven by an aging population and established healthcare systems. However, the Asia Pacific region is projected to be the fastest-growing market, attributed to improving healthcare access, increasing disposable incomes, and a large patient pool in countries like China and India.

