Pirfenidone Market
Pirfenidone Market Size, Scope, Growth, Trends and By Segmentation Types, Applications, Regional Analysis and Industry Forecast (2025-2033)
Report ID : RI_701739 | Last Updated : July 30, 2025 |
Format : ![]()
![]()
![]()
![]()
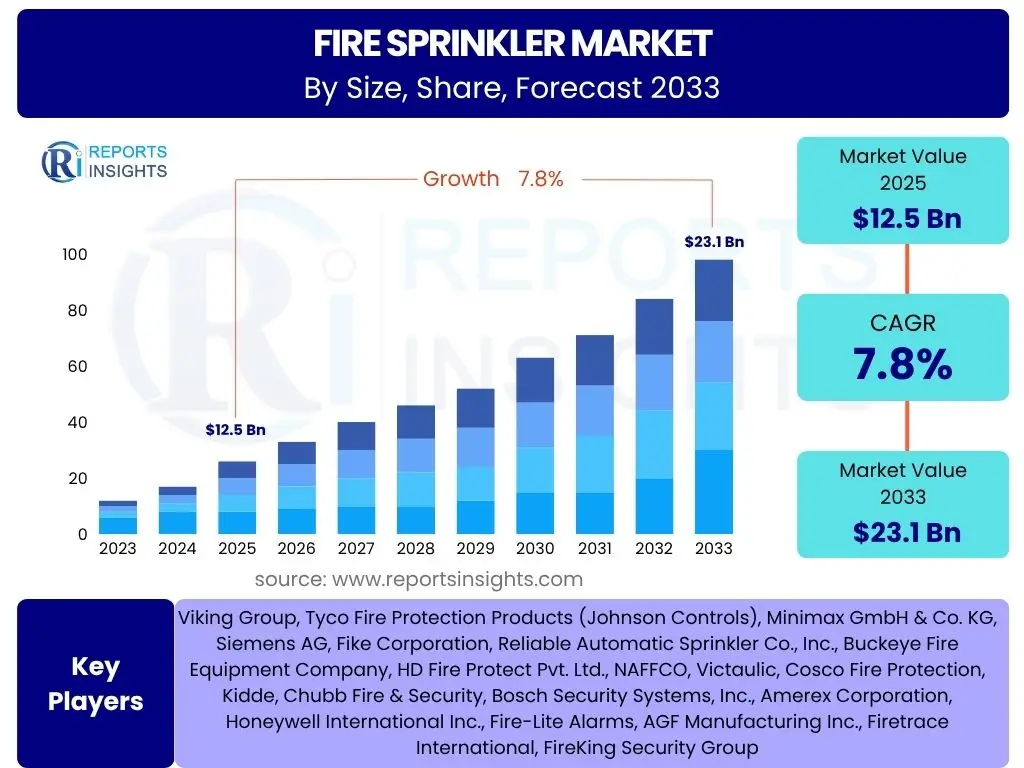
Pirfenidone Market Size
According to Reports Insights Consulting Pvt Ltd, The Pirfenidone Market is projected to grow at a Compound Annual Growth Rate (CAGR) of 6.8% between 2025 and 2033. The market is estimated at USD 1.75 Billion in 2025 and is projected to reach USD 2.95 Billion by the end of the forecast period in 2033.
Key Pirfenidone Market Trends & Insights
The Pirfenidone market is experiencing dynamic shifts driven by evolving therapeutic landscapes and increasing global health awareness regarding fibrotic lung diseases. A significant trend involves the expanding understanding of Idiopathic Pulmonary Fibrosis (IPF) pathogenesis, which continues to underscore the critical role of anti-fibrotic agents like Pirfenidone. Furthermore, growing research into combination therapies seeks to enhance treatment efficacy and improve patient outcomes beyond monotherapy approaches.
Another prominent insight relates to the patent expiration of originator drugs in various regions, leading to the emergence of generic versions of Pirfenidone. This trend is expected to significantly influence market dynamics by improving drug accessibility and affordability, potentially broadening the patient base, particularly in developing economies. Additionally, there is a sustained focus on early diagnosis and intervention strategies for IPF, which, coupled with enhanced diagnostic capabilities, contributes to the demand for effective treatments.
- Increasing prevalence and diagnosis of Idiopathic Pulmonary Fibrosis (IPF) globally.
- Development and adoption of generic Pirfenidone formulations following patent expirations, enhancing accessibility.
- Growing research and clinical trials exploring combination therapies to improve efficacy.
- Rising awareness among healthcare professionals and patients regarding fibrotic lung diseases.
- Advancements in diagnostic technologies facilitating earlier and more accurate IPF detection.
- Expansion of Pirfenidone's approved indications to include other fibrotic conditions.
AI Impact Analysis on Pirfenidone
Artificial Intelligence (AI) is progressively reshaping various facets of pharmaceutical research, development, and patient management within the Pirfenidone domain. Users frequently inquire about how AI can accelerate drug discovery, optimize clinical trials, and personalize patient treatment strategies for fibrotic diseases. AI algorithms are proving instrumental in identifying novel therapeutic targets and predicting drug efficacy, potentially leading to the development of next-generation anti-fibrotic agents or optimized Pirfenidone derivatives.
In the clinical sphere, AI's influence extends to enhancing patient stratification, improving recruitment for clinical trials, and monitoring treatment responses more accurately. By analyzing vast datasets of patient genomics, medical imaging, and electronic health records, AI can identify biomarkers indicative of disease progression or treatment susceptibility, enabling more personalized Pirfenidone regimens. This capability promises to refine patient selection, minimize adverse effects, and maximize therapeutic benefits, thereby revolutionizing the management of conditions treated with Pirfenidone.
- AI-driven drug discovery for novel anti-fibrotic compounds and optimized Pirfenidone analogues.
- Enhanced clinical trial design and patient recruitment through AI-powered predictive analytics.
- Personalized medicine approaches utilizing AI to stratify patients for optimal Pirfenidone therapy.
- Improved diagnostic accuracy and prognostic prediction for fibrotic diseases via AI analysis of medical images and clinical data.
- Real-world evidence generation and post-market surveillance supported by AI to monitor Pirfenidone's effectiveness and safety.
- Accelerated R&D processes, reducing time-to-market for new therapeutic strategies.
Key Takeaways Pirfenidone Market Size & Forecast
The Pirfenidone market is poised for sustained growth, driven primarily by the escalating incidence of Idiopathic Pulmonary Fibrosis and the increasing global geriatric population, which is highly susceptible to the condition. A key insight is the expanding geographic reach of Pirfenidone, with significant growth anticipated in emerging economies as healthcare infrastructure improves and patient access to advanced therapies increases. The market forecast reflects a positive trajectory, underpinned by ongoing clinical research aimed at broadening the therapeutic applications of Pirfenidone and improving its efficacy profile.
Furthermore, the market's trajectory will be significantly influenced by the interplay between originator drugs and their generic counterparts. While patent expirations introduce competitive pricing pressures, they also expand the market by making treatment more affordable and accessible to a wider patient demographic. Strategic partnerships and collaborations among pharmaceutical companies, along with governmental support for rare disease research, are expected to further bolster market expansion and drive innovation in the Pirfenidone landscape.
- Robust growth fueled by the rising global prevalence of Idiopathic Pulmonary Fibrosis (IPF) and an aging population.
- Significant market expansion due to the increasing availability and adoption of generic Pirfenidone versions.
- Growing emphasis on early diagnosis and intervention, supporting increased drug prescriptions.
- Potential for new indications beyond IPF and the exploration of combination therapies to drive future growth.
- North America and Europe currently dominate the market, with Asia Pacific exhibiting the highest growth potential.
Pirfenidone Market Drivers Analysis
The Pirfenidone market is primarily driven by the increasing global prevalence of Idiopathic Pulmonary Fibrosis (IPF), a chronic and progressive lung disease with significant unmet medical needs. The aging global population contributes substantially to this prevalence, as IPF predominantly affects individuals over the age of 60. Concurrently, advancements in diagnostic techniques and rising awareness among healthcare professionals and patients have led to earlier and more accurate diagnoses, expanding the treatable patient pool.
Supportive governmental initiatives and favorable reimbursement policies in key markets also play a crucial role in enhancing patient access to Pirfenidone, mitigating the financial burden of treatment. Furthermore, ongoing research and development efforts aimed at exploring new therapeutic applications for Pirfenidone beyond IPF, as well as the potential for combination therapies, are expected to create additional market opportunities and reinforce its position as a vital anti-fibrotic agent.
| Drivers | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Increasing Prevalence of Idiopathic Pulmonary Fibrosis (IPF) | +1.5% | Global, particularly North America, Europe, Asia Pacific | Short to Long-term (2025-2033) |
| Aging Global Population | +1.2% | Global, particularly developed economies | Short to Long-term (2025-2033) |
| Advancements in Diagnostic Techniques for IPF | +0.8% | Global | Mid-term (2027-2033) |
| Rising Awareness and Patient Education Initiatives | +0.7% | Global, especially emerging markets | Mid-term (2027-2033) |
| Favorable Reimbursement Policies and Government Support | +0.6% | North America, Europe, select Asian countries | Short to Mid-term (2025-2030) |
Pirfenidone Market Restraints Analysis
Despite its therapeutic benefits, the Pirfenidone market faces significant restraints, primarily the high cost of therapy, which can limit patient access, especially in regions with inadequate insurance coverage or healthcare systems. The high price point poses a substantial barrier to widespread adoption, particularly in developing countries, and can lead to patient non-adherence due to financial strain. This economic burden necessitates robust reimbursement policies to ensure equitable access.
Another major restraint involves the side effects associated with Pirfenidone, such as gastrointestinal disturbances and photosensitivity, which can lead to treatment discontinuation in a notable percentage of patients. While these side effects are generally manageable, their occurrence impacts patient quality of life and adherence, thereby affecting the drug's overall market uptake. Furthermore, the availability of alternative treatments for IPF, including other anti-fibrotic drugs, introduces market competition and can restrain Pirfenidone's market share if perceived to offer superior efficacy or a better side-effect profile.
| Restraints | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| High Cost of Therapy | -1.0% | Global, particularly developing nations | Short to Long-term (2025-2033) |
| Associated Side Effects and Patient Non-Adherence | -0.8% | Global | Short to Long-term (2025-2033) |
| Presence of Alternative Therapies for IPF | -0.5% | Global | Short to Long-term (2025-2033) |
| Strict Regulatory Approval Processes in Some Regions | -0.3% | Select countries with stringent regulations | Short to Mid-term (2025-2030) |
Pirfenidone Market Opportunities Analysis
The Pirfenidone market presents several compelling opportunities for growth and expansion. A primary opportunity lies in the exploration and expansion into new indications beyond Idiopathic Pulmonary Fibrosis (IPF). Research into Pirfenidone's anti-fibrotic properties suggests potential therapeutic applications in other fibrotic diseases affecting organs such as the kidney, liver, and skin, which could significantly broaden its market scope and patient population. This diversification of indications would open new revenue streams and address additional unmet medical needs.
Another significant opportunity stems from the development of combination therapies. Investigating Pirfenidone's synergy with other existing or novel compounds could lead to enhanced efficacy, reduced side effects, or improved patient outcomes compared to monotherapy. This approach holds promise for more aggressive disease management and positions Pirfenidone as a foundational component in multi-drug regimens. Furthermore, the growing healthcare infrastructure and increasing affordability of generic versions in emerging markets represent a substantial untapped potential, allowing for greater market penetration and patient access in previously underserved regions.
| Opportunities | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Expansion into New Indications (e.g., other fibrotic diseases) | +1.3% | Global | Mid to Long-term (2028-2033) |
| Development of Combination Therapies | +1.0% | Global | Mid to Long-term (2027-2033) |
| Growth in Emerging Markets Due to Increased Access | +0.9% | Asia Pacific, Latin America, MEA | Short to Long-term (2025-2033) |
| Strategic Collaborations and Partnerships | +0.5% | Global | Short to Mid-term (2025-2030) |
Pirfenidone Market Challenges Impact Analysis
The Pirfenidone market faces several inherent challenges that could impede its growth trajectory. A major hurdle is the limited efficacy of Pirfenidone in reversing established fibrosis or curing IPF, meaning it primarily slows disease progression rather than stopping or reversing it. This limitation means patients still face progressive decline, which can influence treatment adherence and the perceived value of the therapy. Further research is ongoing to address this unmet need, but it remains a significant challenge.
Moreover, patent expirations for innovator Pirfenidone products have led to a surge in generic versions. While this increases accessibility, it also intensifies price competition, potentially eroding the market value and profitability for originator companies. This competitive pressure necessitates strategic pricing and differentiated value propositions. Additionally, the development of novel and potentially more effective anti-fibrotic agents by competitors could pose a long-term threat, requiring continuous innovation and clinical evidence generation to maintain Pirfenidone's market relevance.
| Challenges | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Limited Efficacy in Reversing Fibrosis and Overall Cure | -0.7% | Global | Short to Long-term (2025-2033) |
| Intense Price Competition from Generic Versions | -0.9% | Global, particularly developed markets | Short to Mid-term (2025-2030) |
| Development of Potentially Superior Alternative Therapies | -0.6% | Global | Mid to Long-term (2028-2033) |
| Patient Adherence and Compliance Issues | -0.4% | Global | Short to Long-term (2025-2033) |
Pirfenidone Market - Updated Report Scope
This report provides an in-depth analysis of the global Pirfenidone market, encompassing market size estimations, growth drivers, restraints, opportunities, and challenges across various segments and key geographical regions. It offers a comprehensive overview of the competitive landscape, highlighting the strategies of leading market players and the impact of emerging trends. The report aims to furnish stakeholders with critical insights to inform strategic decisions and navigate the evolving market dynamics of anti-fibrotic therapies.
| Report Attributes | Report Details |
|---|---|
| Base Year | 2024 |
| Historical Year | 2019 to 2023 |
| Forecast Year | 2025 - 2033 |
| Market Size in 2025 | USD 1.75 Billion |
| Market Forecast in 2033 | USD 2.95 Billion |
| Growth Rate | 6.8% |
| Number of Pages | 257 |
| Key Trends |
|
| Segments Covered |
|
| Key Companies Covered | Roche AG, Cipla Inc., Lupin Limited, Dr. Reddy's Laboratories Ltd., Sun Pharmaceutical Industries Ltd., Mylan N.V. (Viatris Inc.), Accord Healthcare, Zentiva Group, Apotex Inc., Hikma Pharmaceuticals PLC, Hetero Drugs Limited, Jubilant Life Sciences Limited, Cadila Pharmaceuticals, Glenmark Pharmaceuticals Limited, Teva Pharmaceutical Industries Ltd., Alkem Laboratories Ltd., Torrent Pharmaceuticals Ltd., Aurobindo Pharma Ltd., Zydus Cadila, Natco Pharma Ltd. |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Speak to Analyst | Avail customised purchase options to meet your exact research needs. Request For Analyst Or Customization |
Segmentation Analysis
The Pirfenidone market is meticulously segmented to provide a granular understanding of its diverse components and drivers. This segmentation allows for precise market analysis, identifying distinct demand patterns and growth opportunities across different therapeutic areas, product forms, and distribution channels. Understanding these segments is crucial for stakeholders to develop targeted strategies and optimize resource allocation within the complex pharmaceutical landscape.
Key segmentation categories include the primary application areas for Pirfenidone, such as its established use in Idiopathic Pulmonary Fibrosis (IPF) and emerging potential in other fibrotic conditions, reflecting the drug's expanding therapeutic utility. Further segmentation by dosage form (oral tablets and capsules) addresses patient preference and administration convenience. The market is also analyzed based on distribution channels, including hospital, retail, and online pharmacies, providing insights into procurement and access dynamics. Finally, end-user segmentation examines the adoption across various healthcare settings like hospitals, clinics, and homecare, indicating where the drug is most frequently dispensed and administered.
- By Application
- Idiopathic Pulmonary Fibrosis (IPF)
- Other Fibrotic Conditions (e.g., Scleroderma-associated Interstitial Lung Disease, Chronic Hypersensitivity Pneumonitis)
- By Dosage Form
- Oral Tablets
- Oral Capsules
- By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Specialty Pharmacies
- By End-User
- Hospitals
- Clinics
- Homecare Settings
- Research Institutes
Regional Highlights
- North America: This region holds the largest market share for Pirfenidone, primarily driven by the high prevalence of IPF, well-established healthcare infrastructure, advanced diagnostic capabilities, and favorable reimbursement policies. The presence of key market players and a strong focus on research and development also contribute significantly to its dominance.
- Europe: Europe represents a substantial market for Pirfenidone, characterized by an aging population susceptible to IPF, increasing awareness of fibrotic lung diseases, and supportive government initiatives for orphan drugs. Regulatory approvals and a robust healthcare system further bolster market growth in this region.
- Asia Pacific (APAC): The Asia Pacific region is projected to exhibit the highest growth rate during the forecast period. This growth is attributed to a large and expanding patient pool, improving healthcare accessibility, increasing healthcare expenditure, and the rising availability of generic Pirfenidone versions. Countries like China, India, and Japan are key contributors to this growth.
- Latin America: This region is experiencing steady growth, driven by increasing awareness of IPF, improving healthcare infrastructure, and a gradual expansion of access to specialized treatments. Economic development and healthcare reforms are expected to further support market expansion.
- Middle East and Africa (MEA): The MEA region is an emerging market for Pirfenidone, with growth propelled by rising healthcare investments, increasing prevalence of chronic diseases including IPF, and efforts to enhance access to advanced therapies. However, market growth may be slower due to varying healthcare standards and economic disparities.
Top Key Players
The market research report includes a detailed profile of leading stakeholders in the Pirfenidone Market.- Roche AG
- Cipla Inc.
- Lupin Limited
- Dr. Reddy's Laboratories Ltd.
- Sun Pharmaceutical Industries Ltd.
- Mylan N.V. (Viatris Inc.)
- Accord Healthcare
- Zentiva Group
- Apotex Inc.
- Hikma Pharmaceuticals PLC
- Hetero Drugs Limited
- Jubilant Life Sciences Limited
- Cadila Pharmaceuticals
- Glenmark Pharmaceuticals Limited
- Teva Pharmaceutical Industries Ltd.
- Alkem Laboratories Ltd.
- Torrent Pharmaceuticals Ltd.
- Aurobindo Pharma Ltd.
- Zydus Cadila
- Natco Pharma Ltd.
Frequently Asked Questions
Analyze common user questions about the Pirfenidone market and generate a concise list of summarized FAQs reflecting key topics and concerns.What is Pirfenidone primarily used for?
Pirfenidone is primarily used for the treatment of Idiopathic Pulmonary Fibrosis (IPF), a chronic, progressive, and often fatal lung disease characterized by the scarring of lung tissue. It helps to slow the progression of the disease and preserve lung function.
How does Pirfenidone work?
Pirfenidone is an anti-fibrotic agent that works by reducing the production of growth factors and pro-collagen, which are involved in the development of fibrosis or scarring in the lungs. Its exact mechanism is not fully understood but involves anti-inflammatory and antioxidant properties.
What are the common side effects of Pirfenidone?
Common side effects of Pirfenidone include gastrointestinal issues such as nausea, diarrhea, dyspepsia, and abdominal pain. Other side effects can include skin reactions like rash and photosensitivity, fatigue, and headaches. Patients are often advised to take it with food to minimize gastrointestinal discomfort.
Is Pirfenidone a cure for Idiopathic Pulmonary Fibrosis (IPF)?
No, Pirfenidone is not a cure for Idiopathic Pulmonary Fibrosis (IPF). It is an anti-fibrotic medication that helps to slow the rate of decline in lung function and disease progression, but it does not reverse the existing lung damage or offer a definitive cure for the condition.
What is the market outlook for Pirfenidone?
The Pirfenidone market is projected for sustained growth, driven by the increasing incidence of IPF, an aging global population, and advancements in early diagnosis. The market is also benefiting from the increased availability of more affordable generic versions and the potential for expanded therapeutic indications.

