Pharmaceutical Grade Sorbitan Ester Market
Pharmaceutical Grade Sorbitan Ester Market Size, Scope, Growth, Trends and By Segmentation Types, Applications, Regional Analysis and Industry Forecast (2025-2033)
Report ID : RI_701132 | Last Updated : July 29, 2025 |
Format : ![]()
![]()
![]()
![]()
Pharmaceutical Grade Sorbitan Ester Market Size
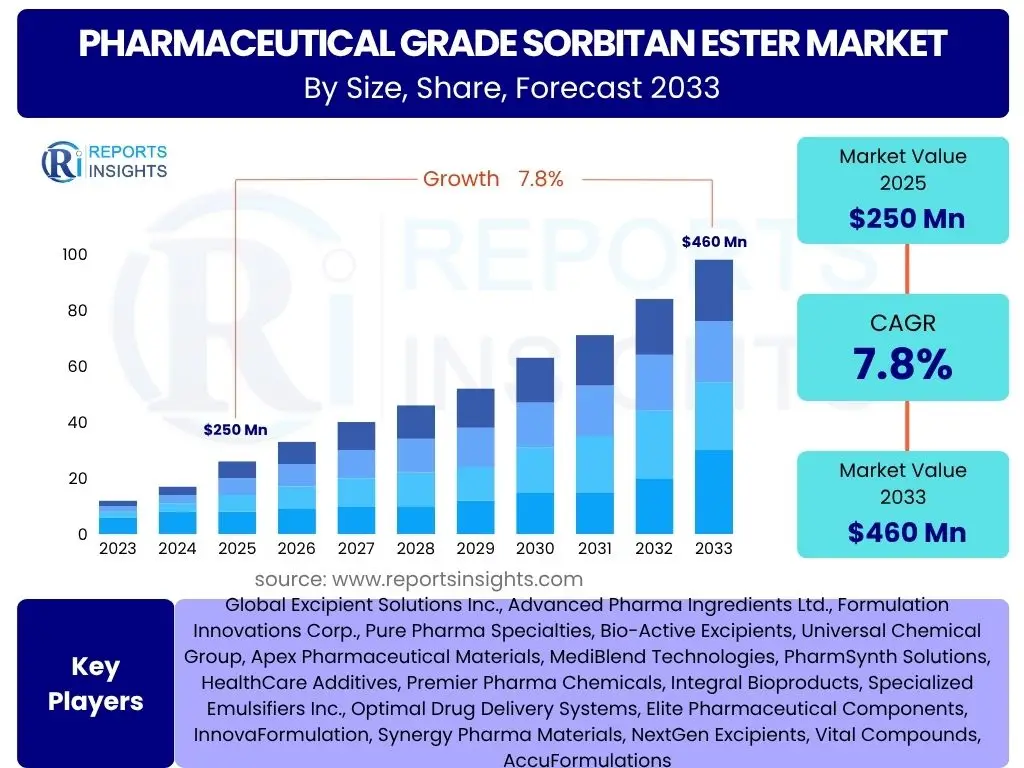
According to Reports Insights Consulting Pvt Ltd, The Pharmaceutical Grade Sorbitan Ester Market is projected to grow at a Compound Annual Growth Rate (CAGR) of 7.8% between 2025 and 2033. The market is estimated at USD 250 Million in 2025 and is projected to reach USD 460 Million by the end of the forecast period in 2033.
Key Pharmaceutical Grade Sorbitan Ester Market Trends & Insights
The Pharmaceutical Grade Sorbitan Ester market is witnessing significant transformation driven by evolving pharmaceutical formulation needs and increasing demand for high-purity, multifunctional excipients. Key trends indicate a pronounced shift towards specialized drug delivery systems, where sorbitan esters play a critical role as emulsifiers, solubilizers, and stabilizers. This includes the growing adoption in parenteral and ophthalmic formulations, where their non-ionic and biocompatible properties are highly valued for enhancing drug solubility and stability. Furthermore, there is an increasing emphasis on sustainable and naturally derived ingredients, prompting manufacturers to explore bio-based sorbitan ester production methods to meet the rising demand for eco-friendly pharmaceutical components.
Another prominent trend is the rising demand for sophisticated excipients that can improve the bioavailability of poorly soluble drugs. Sorbitan esters are proving instrumental in lipid-based drug delivery systems and nanoemulsions, addressing solubility challenges and enabling more effective therapeutic outcomes. The expanding biopharmaceutical sector, particularly in the development of protein and peptide drugs, also contributes to this trend, as these complex molecules often require precise stabilization and solubilization that sorbitan esters can provide. The market is also experiencing a surge in customization, with pharmaceutical companies seeking tailor-made sorbitan ester blends to optimize specific drug formulations, necessitating close collaboration between excipient manufacturers and drug developers.
- Increasing adoption in advanced drug delivery systems such as nanoemulsions and lipid-based formulations.
- Growing demand for high-purity and specialized sorbitan esters for sensitive pharmaceutical applications.
- Shift towards bio-based and sustainable excipient manufacturing practices.
- Rising use in parenteral and ophthalmic drug formulations due to excellent biocompatibility.
- Expanding application in the biopharmaceutical sector for protein and peptide stabilization.
- Customization of sorbitan ester blends to meet specific formulation requirements.
AI Impact Analysis on Pharmaceutical Grade Sorbitan Ester
The integration of Artificial intelligence (AI) is set to revolutionize various facets of the pharmaceutical industry, and its impact on the Pharmaceutical Grade Sorbitan Ester market, while indirect, is significant. AI can optimize the discovery and development of novel drug formulations by predicting excipient compatibility, stability, and efficacy. Through advanced algorithms, researchers can rapidly screen and select the most suitable sorbitan ester types and concentrations for a given active pharmaceutical ingredient (API), significantly accelerating pre-formulation studies and reducing experimental costs. This predictive capability extends to analyzing complex interactions between APIs and excipients, ensuring optimal drug delivery and bioavailability profiles, ultimately driving demand for specific, high-performance sorbitan esters.
Beyond R&D, AI’s influence extends to manufacturing and supply chain management within the excipient industry. AI-powered predictive maintenance can optimize production processes for sorbitan esters, ensuring consistent quality, reducing waste, and improving operational efficiency. Furthermore, AI algorithms can enhance supply chain resilience by forecasting demand fluctuations, optimizing inventory levels, and identifying potential disruptions in raw material sourcing for sorbitan ester production. This leads to more stable supply and pricing for pharmaceutical manufacturers, indirectly supporting the wider adoption of these critical excipients. Ethical considerations and data privacy related to AI applications in pharmaceutical data, however, remain a key area of focus for industry stakeholders to ensure responsible innovation.
- Accelerated formulation development and excipient selection through AI-driven predictive modeling.
- Optimized manufacturing processes for sorbitan esters, leading to improved consistency and yield.
- Enhanced quality control and impurity detection using AI-powered analytical tools.
- Improved supply chain efficiency and resilience for raw materials and finished sorbitan esters.
- Potential for AI to identify novel applications or modifications of sorbitan esters for complex drugs.
Key Takeaways Pharmaceutical Grade Sorbitan Ester Market Size & Forecast
The Pharmaceutical Grade Sorbitan Ester market is poised for robust expansion, driven primarily by the escalating demand for advanced drug delivery systems and the continuous innovation in pharmaceutical formulations. The forecast period indicates a steady growth trajectory, underpinned by the increasing prevalence of chronic diseases necessitating effective drug administration and the expanding pipeline of complex biopharmaceuticals. A key takeaway is the critical role these excipients play in enhancing drug solubility, stability, and bioavailability, making them indispensable components in modern drug development and manufacturing processes.
Another crucial insight from the market size and forecast is the strategic importance of regional pharmaceutical hubs, particularly in North America, Europe, and the Asia-Pacific, which are driving significant demand due to high R&D investments and robust healthcare infrastructure. Manufacturers are increasingly focusing on product purity, regulatory compliance, and functional versatility to meet the stringent requirements of the pharmaceutical industry. The market's future will largely depend on sustained innovation in excipient technology, strategic collaborations between ingredient suppliers and drug developers, and the ability to navigate evolving regulatory landscapes globally.
- The market exhibits strong growth potential, projected to reach USD 460 Million by 2033.
- Demand is significantly influenced by advancements in drug delivery technologies and biopharmaceutical development.
- High purity and functional versatility are critical attributes driving product adoption.
- North America, Europe, and Asia Pacific are expected to remain key revenue generators.
- Regulatory adherence and consistent product quality are paramount for market players.
Pharmaceutical Grade Sorbitan Ester Market Drivers Analysis
The Pharmaceutical Grade Sorbitan Ester market is propelled by several robust drivers, fundamentally rooted in the evolving landscape of drug development and patient care. A primary driver is the surging demand for advanced drug delivery systems, such as nanoemulsions, microemulsions, and lipid-based formulations. Sorbitan esters, with their excellent emulsifying and stabilizing properties, are crucial in these systems for enhancing the solubility, bioavailability, and targeted delivery of poorly soluble active pharmaceutical ingredients (APIs). This innovation in drug delivery directly translates into a higher consumption of these specialized excipients. Furthermore, the global rise in chronic diseases, including cardiovascular conditions, diabetes, and various cancers, necessitates the development of more effective and patient-compliant drug therapies, often relying on sophisticated formulations that incorporate sorbitan esters.
Another significant driver is the rapid growth of the biopharmaceutical sector, particularly the development of complex protein and peptide therapeutics. These biological molecules are often delicate and prone to degradation, requiring highly stable and biocompatible excipients to maintain their structural integrity and efficacy. Sorbitan esters serve as vital stabilizers and solubilizers in these formulations, preventing aggregation and improving the shelf-life of biologics. Additionally, increasing pharmaceutical research and development (R&D) investments globally, aimed at discovering new drugs and reformulating existing ones for improved performance, contribute substantially to the demand for high-quality pharmaceutical grade sorbitan esters. The expanding geriatric population, with its associated increase in demand for diverse pharmaceutical products, further underscores the necessity for these versatile excipients.
| Drivers | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Growing Demand for Advanced Drug Delivery Systems | +2.1% | Global, North America, Europe, Asia Pacific | 2025-2033 |
| Rising R&D Investments in Pharmaceutical Formulations | +1.8% | Global, USA, Germany, Japan, China | 2025-2033 |
| Expansion of the Biopharmaceutical Sector | +1.7% | Global, especially developed economies | 2025-2033 |
| Increasing Prevalence of Chronic Diseases | +1.5% | Global | 2025-2033 |
| Advancements in Emulsification and Solubilization Technologies | +0.7% | Global | 2025-2033 |
Pharmaceutical Grade Sorbitan Ester Market Restraints Analysis
Despite robust growth drivers, the Pharmaceutical Grade Sorbitan Ester market faces several restraints that could impede its full potential. One significant restraint is the stringent regulatory approval processes governing pharmaceutical excipients. Manufacturers must adhere to rigorous quality standards, purity requirements, and documentation protocols, such as Good Manufacturing Practices (GMP) and pharmacopoeial specifications, which can lead to high compliance costs and lengthy approval timelines. This regulatory burden can particularly challenge new entrants or smaller manufacturers, limiting market diversification and innovation. Any failure to meet these strict guidelines can result in product recalls or market exclusion, impacting market growth and trust.
Another key restraint is the availability of alternative excipients and the potential for substitution. While sorbitan esters offer unique properties, other emulsifiers, solubilizers, and stabilizers, such as polysorbates, phospholipids, or polyethylene glycols (PEGs), may sometimes offer comparable functionality or cost advantages in certain formulations. The decision to use a specific excipient is often driven by a complex interplay of cost, performance, regulatory acceptance, and formulation-specific needs. Furthermore, fluctuations in raw material prices, primarily sorbitol and fatty acids, can impact the production cost of sorbitan esters, affecting pricing strategies and profit margins for manufacturers. Supply chain vulnerabilities and the need for consistent, high-quality raw materials also pose ongoing challenges.
| Restraints | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Stringent Regulatory Approval Processes | -1.2% | Global, highly regulated markets (e.g., US, EU) | 2025-2033 |
| Availability of Alternative Excipients | -0.8% | Global | 2025-2033 |
| Fluctuations in Raw Material Prices | -0.6% | Global | 2025-2033 |
| High Production and R&D Costs for Special Grades | -0.5% | Global | 2025-2033 |
Pharmaceutical Grade Sorbitan Ester Market Opportunities Analysis
Significant opportunities exist within the Pharmaceutical Grade Sorbitan Ester market, particularly driven by the increasing focus on specialty and high-purity excipients. There is a growing demand for tailor-made sorbitan ester solutions that address specific challenges in complex drug formulations, such as those involving highly potent APIs or novel drug delivery platforms. This creates avenues for manufacturers to invest in advanced research and development, offering customized products that provide enhanced functionality, improved stability, and superior compatibility. The expansion of pharmaceutical manufacturing capabilities in emerging economies, particularly in Asia Pacific and Latin America, also presents lucrative growth prospects as these regions witness increasing healthcare expenditure and a burgeoning generic drug industry, which requires high-quality excipients.
Furthermore, the shift towards non-invasive drug delivery methods, including transdermal patches, nasal sprays, and pulmonary inhalers, offers new application areas for sorbitan esters. Their role in enhancing drug permeation and formulation stability in these delivery systems is being increasingly recognized, opening up novel market segments. Strategic collaborations and partnerships between excipient manufacturers and pharmaceutical companies can also unlock significant opportunities by fostering innovation, streamlining supply chains, and accelerating product development cycles. Investment in sustainable production technologies and the development of bio-based sorbitan esters align with global environmental trends and consumer preferences, potentially offering a competitive edge and unlocking new market segments focusing on green pharmaceutical ingredients.
| Opportunities | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Expansion into Emerging Pharmaceutical Markets | +1.5% | Asia Pacific, Latin America, MEA | 2025-2033 |
| Growing Demand for Specialty and Customized Excipients | +1.3% | Global | 2025-2033 |
| Technological Advancements in Non-Invasive Drug Delivery | +1.1% | Global | 2025-2033 |
| Strategic Collaborations and Partnerships | +0.9% | Global | 2025-2033 |
| Development of Bio-based and Sustainable Sorbitan Esters | +0.7% | Europe, North America | 2025-2033 |
Pharmaceutical Grade Sorbitan Ester Market Challenges Impact Analysis
The Pharmaceutical Grade Sorbitan Ester market faces several enduring challenges that require strategic navigation from industry participants. A primary challenge involves maintaining consistent product quality and purity across varied manufacturing batches, especially given the strict requirements of pharmaceutical applications. Any slight variation in impurities or physiochemical properties can impact drug stability, efficacy, or patient safety, leading to costly recalls or regulatory non-compliance. This necessitates significant investment in advanced analytical techniques, rigorous quality control protocols, and robust supply chain management to ensure the integrity of raw materials and final products, adding complexity and cost to operations.
Another significant challenge is the intense competition and pricing pressure within the excipient market. While demand for high-quality sorbitan esters is growing, the market also features numerous players, leading to competitive pricing that can squeeze profit margins, particularly for standardized grades. Furthermore, geopolitical instabilities, trade wars, and global health crises can disrupt supply chains for critical raw materials, leading to shortages or price volatility. Navigating complex intellectual property rights, ensuring a skilled workforce, and adapting to rapidly evolving environmental regulations, such as those pertaining to manufacturing emissions and waste disposal, also present ongoing hurdles for market players aiming for sustainable growth and long-term viability.
| Challenges | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Maintaining Consistent High Product Quality and Purity | -1.0% | Global | 2025-2033 |
| Intense Competition and Pricing Pressure | -0.7% | Global | 2025-2033 |
| Supply Chain Disruptions and Raw Material Volatility | -0.6% | Global | 2025-2033 |
| Intellectual Property Protection and Innovation Imitation | -0.4% | Global | 2025-2033 |
Pharmaceutical Grade Sorbitan Ester Market - Updated Report Scope
This market research report provides a comprehensive analysis of the Pharmaceutical Grade Sorbitan Ester market, offering detailed insights into market dynamics, segmentation, regional trends, and competitive landscape. The scope encompasses a thorough examination of market size, growth forecasts, key drivers, restraints, opportunities, and challenges influencing the industry's trajectory. It further delves into the impact of emerging technologies like AI and highlights critical industry trends shaping the future of pharmaceutical excipients. The report aims to equip stakeholders with actionable intelligence for strategic decision-making and market positioning.
| Report Attributes | Report Details |
|---|---|
| Base Year | 2024 |
| Historical Year | 2019 to 2023 |
| Forecast Year | 2025 - 2033 |
| Market Size in 2025 | USD 250 Million |
| Market Forecast in 2033 | USD 460 Million |
| Growth Rate | 7.8% |
| Number of Pages | 245 |
| Key Trends |
|
| Segments Covered |
|
| Key Companies Covered | Global Excipient Solutions Inc., Advanced Pharma Ingredients Ltd., Formulation Innovations Corp., Pure Pharma Specialties, Bio-Active Excipients, Universal Chemical Group, Apex Pharmaceutical Materials, MediBlend Technologies, PharmSynth Solutions, HealthCare Additives, Premier Pharma Chemicals, Integral Bioproducts, Specialized Emulsifiers Inc., Optimal Drug Delivery Systems, Elite Pharmaceutical Components, InnovaFormulation, Synergy Pharma Materials, NextGen Excipients, Vital Compounds, AccuFormulations |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Speak to Analyst | Avail customised purchase options to meet your exact research needs. Request For Analyst Or Customization |
Segmentation Analysis
The Pharmaceutical Grade Sorbitan Ester market is comprehensively segmented to provide a granular view of its diverse applications and product types, enabling a detailed understanding of market dynamics. This segmentation facilitates analysis of specific product demands, end-user preferences, and regional consumption patterns, crucial for strategic planning. The market is primarily categorized by type, application, and end-use, reflecting the varied roles sorbitan esters play across the pharmaceutical industry.
The segmentation by type distinguishes between different ester forms, each possessing unique properties suitable for specific formulation requirements. Application segmentation highlights the broad range of pharmaceutical products that incorporate these excipients, from traditional oral medications to highly specialized parenteral and ophthalmic preparations. Furthermore, the end-use segmentation provides insight into the primary consumers of pharmaceutical grade sorbitan esters, including pharmaceutical manufacturers, contract research organizations, and biotechnology firms, underscoring the industry's integrated value chain.
- By Type:
- Sorbitan Monostearate (SMS): Widely used as an emulsifier and stabilizer, particularly in topical creams, ointments, and oral suspensions.
- Sorbitan Monooleate (SMO): Known for its emulsifying properties, often utilized in parenteral and veterinary preparations.
- Sorbitan Monolaurate (SML): Employed as a wetting agent and emulsifier in various pharmaceutical formulations.
- Sorbitan Sesquioleate (SSO): Offers strong emulsifying capabilities, suitable for water-in-oil emulsions.
- Sorbitan Trioleate (STO): A lipophilic emulsifier often used in specialized drug delivery systems.
- Others: Includes less common or custom-blended sorbitan esters for niche applications.
- By Application:
- Oral Formulations: Tablets, capsules, suspensions, and syrups.
- Topical Formulations: Creams, ointments, lotions, and transdermal patches.
- Parenteral Formulations: Injections and sterile solutions requiring high purity.
- Ophthalmic Formulations: Eye drops and ointments where biocompatibility is crucial.
- Veterinary Medicine: Formulations for animal health products.
- Nutraceuticals: Supplements requiring emulsification or solubilization.
- Others: Includes nasal sprays, pulmonary inhalers, and suppositories.
- By End-use:
- Pharmaceutical Companies: Major consumers for their in-house drug manufacturing.
- Contract Manufacturing Organizations (CMOs): Provide formulation and manufacturing services for other pharma companies.
- Research & Academic Institutions: Utilize sorbitan esters for drug discovery, development, and academic studies.
- Biotech Companies: For developing and manufacturing biologics and advanced therapies.
Regional Highlights
- North America: The North American market, particularly the United States and Canada, represents a significant share of the Pharmaceutical Grade Sorbitan Ester market. This dominance is attributed to a robust pharmaceutical industry with extensive R&D investments, a high prevalence of chronic diseases, and a strong focus on advanced drug delivery systems. The presence of leading pharmaceutical and biotechnology companies, coupled with stringent regulatory standards and a well-established healthcare infrastructure, drives the demand for high-purity and specialized excipients. Continuous innovation in drug formulation and a growing demand for biologics further bolster market growth in this region.
- Europe: Europe is another major contributor to the global market, driven by its well-developed pharmaceutical sector, increasing geriatric population, and a strong emphasis on research and development. Countries like Germany, France, and the UK are at the forefront of pharmaceutical innovation, fostering demand for advanced excipients like sorbitan esters in novel drug formulations. The region's adherence to Good Manufacturing Practices (GMP) and stringent quality control further ensures the demand for premium-grade ingredients. The expansion of biopharmaceutical production and rising investments in personalized medicine also contribute significantly to market expansion across the continent.
- Asia Pacific (APAC): The Asia Pacific region is projected to be the fastest-growing market for Pharmaceutical Grade Sorbitan Esters, primarily due to the rapid expansion of the pharmaceutical industry in countries like China, India, and Japan. This growth is fueled by increasing healthcare expenditure, a large patient pool, and government initiatives promoting domestic pharmaceutical manufacturing and R&D. The rising demand for generic drugs and the growing adoption of advanced drug delivery technologies are key drivers. Furthermore, the region is becoming a hub for contract manufacturing and research organizations, which are significant consumers of pharmaceutical excipients.
- Latin America: The Latin American market for Pharmaceutical Grade Sorbitan Esters is experiencing steady growth, driven by improving healthcare infrastructure, increasing access to medicines, and the expansion of local pharmaceutical production. Countries such as Brazil and Mexico are leading the regional market, supported by a growing demand for both branded and generic drugs. Investment in pharmaceutical R&D, albeit at a slower pace compared to developed regions, is gradually increasing, creating new opportunities for excipient manufacturers. Economic development and a rising awareness of advanced healthcare solutions are also contributing factors.
- Middle East and Africa (MEA): The MEA region is an emerging market for Pharmaceutical Grade Sorbitan Esters, characterized by developing healthcare systems and growing pharmaceutical investments, particularly in countries like Saudi Arabia, UAE, and South Africa. While currently a smaller market share, the region presents future growth opportunities due to rising health expenditure, increasing prevalence of non-communicable diseases, and efforts to reduce reliance on imported pharmaceuticals through local manufacturing. Regulatory reforms aimed at improving pharmaceutical quality and safety also support the demand for high-grade excipients in the region.
Top Key Players
The market research report includes a detailed profile of leading stakeholders in the Pharmaceutical Grade Sorbitan Ester Market.- Global Excipient Solutions Inc.
- Advanced Pharma Ingredients Ltd.
- Formulation Innovations Corp.
- Pure Pharma Specialties
- Bio-Active Excipients
- Universal Chemical Group
- Apex Pharmaceutical Materials
- MediBlend Technologies
- PharmSynth Solutions
- HealthCare Additives
- Premier Pharma Chemicals
- Integral Bioproducts
- Specialized Emulsifiers Inc.
- Optimal Drug Delivery Systems
- Elite Pharmaceutical Components
- InnovaFormulation
- Synergy Pharma Materials
- NextGen Excipients
- Vital Compounds
- AccuFormulations
Frequently Asked Questions
What are pharmaceutical grade sorbitan esters?
Pharmaceutical grade sorbitan esters are a class of non-ionic surfactants used as excipients in drug formulations. They are derivatives of sorbitol (a sugar alcohol) and fatty acids, produced through esterification. These compounds are highly valued in the pharmaceutical industry due to their excellent emulsifying, solubilizing, and stabilizing properties, making them indispensable for various drug delivery systems. Their non-toxic and biocompatible nature ensures their suitability for human consumption and topical applications.
These esters are crucial for creating stable emulsions and dispersions, improving the bioavailability of poorly soluble drugs, and enhancing the overall stability and shelf-life of pharmaceutical products. Different types, such as sorbitan monostearate, monooleate, and monolaurate, offer varying hydrophilic-lipophilic balance (HLB) values, allowing formulators to select the most appropriate ester for specific drug characteristics and intended routes of administration. This versatility underscores their widespread application across oral, topical, parenteral, and ophthalmic formulations, ensuring safe and effective drug delivery.
What drives the growth of the Pharmaceutical Grade Sorbitan Ester market?
The growth of the Pharmaceutical Grade Sorbitan Ester market is primarily driven by the escalating demand for advanced drug delivery systems. Innovations in pharmaceutical formulation, such as nanoemulsions and lipid-based systems, heavily rely on sorbitan esters to enhance drug solubility, stability, and bioavailability, particularly for challenging active pharmaceutical ingredients (APIs). These advanced systems are critical for improving therapeutic outcomes and patient compliance across a spectrum of diseases.
Furthermore, the rapid expansion of the biopharmaceutical sector significantly contributes to market growth. As more complex protein and peptide drugs enter the market, there is a heightened need for highly pure and effective excipients to prevent aggregation and maintain the integrity of these sensitive biologics. Increased global investment in pharmaceutical research and development, aimed at discovering and reformulating drugs, also fuels the demand for high-quality sorbitan esters. The rising prevalence of chronic diseases worldwide further necessitates continuous innovation in drug therapies, sustaining the market's upward trajectory.
What are the main applications of sorbitan esters in pharmaceuticals?
Pharmaceutical grade sorbitan esters find extensive applications across a wide range of drug formulations due to their multifunctional properties. In oral formulations, they serve as emulsifiers and solubilizers in suspensions, syrups, and soft-gel capsules, improving the dissolution and absorption of active pharmaceutical ingredients. For topical applications, such as creams, ointments, and lotions, they act as key emulsifiers and stabilizers, ensuring uniform drug distribution and enhancing skin penetration.
Their high purity and low toxicity make them critical components in parenteral (injectable) and ophthalmic (eye care) formulations, where sterility and biocompatibility are paramount. In these sensitive applications, sorbitan esters help to stabilize emulsions, prevent particle aggregation, and maintain the integrity of sterile solutions. Beyond these, they are increasingly used in advanced drug delivery systems, including lipid-based formulations and nanoemulsions, for poorly soluble drugs, and play a vital role in the stability of protein and peptide therapeutics within the growing biopharmaceutical sector, expanding their utility into specialized and high-value drug products.
Which regions are key contributors to the Pharmaceutical Grade Sorbitan Ester market?
North America and Europe are currently the dominant regions in the Pharmaceutical Grade Sorbitan Ester market, driven by their mature pharmaceutical industries, significant research and development investments, and stringent regulatory environments that necessitate high-quality excipients. The presence of numerous leading pharmaceutical and biotechnology companies, coupled with advanced healthcare infrastructures and a high adoption rate of sophisticated drug delivery technologies, underpins their substantial market shares. These regions continue to lead in innovation and the development of new drug formulations, consistently driving demand for specialized sorbitan esters.
The Asia Pacific (APAC) region, however, is emerging as the fastest-growing market, primarily fueled by the rapid expansion of its pharmaceutical sector in countries like China, India, and Japan. This growth is propelled by increasing healthcare expenditure, a large and aging population, and government initiatives supporting domestic drug manufacturing and R&D. Latin America and the Middle East & Africa (MEA) are also showing promising growth, albeit from a smaller base, as healthcare infrastructure improves and pharmaceutical production expands in these developing economies. Overall, global market growth is influenced by the interplay of established markets maintaining high demand and emerging markets exhibiting rapid expansion.
What challenges does the Pharmaceutical Grade Sorbitan Ester market face?
The Pharmaceutical Grade Sorbitan Ester market encounters several challenges, primarily centered around stringent regulatory compliance and maintaining consistent product quality. Pharmaceutical excipients must adhere to rigorous global pharmacopoeial standards and Good Manufacturing Practices (GMP), which demand significant investment in quality control, testing, and documentation. Non-compliance can lead to costly product recalls or market exclusion, posing a substantial barrier for manufacturers and hindering market entry for new players.
Another significant challenge is the intense market competition and the availability of alternative excipients, which can lead to pricing pressures and margin erosion. While sorbitan esters offer unique benefits, formulators may consider other emulsifiers or solubilizers based on cost, specific formulation needs, or supplier relationships. Furthermore, fluctuations in the prices of raw materials (sorbitol and fatty acids) and potential supply chain disruptions, especially in a globalized market, can impact production costs and stability. Navigating intellectual property rights and ensuring a skilled workforce capable of advanced manufacturing and quality assurance also remain ongoing hurdles for sustainable growth in this specialized market.

