Hemophilia Treatment Drug Market
Hemophilia Treatment Drug Market Size, Scope, Growth, Trends and By Segmentation Types, Applications, Regional Analysis and Industry Forecast (2025-2033)
Report ID : RI_701138 | Last Updated : July 29, 2025 |
Format : ![]()
![]()
![]()
![]()
Hemophilia Treatment Drug Market Size
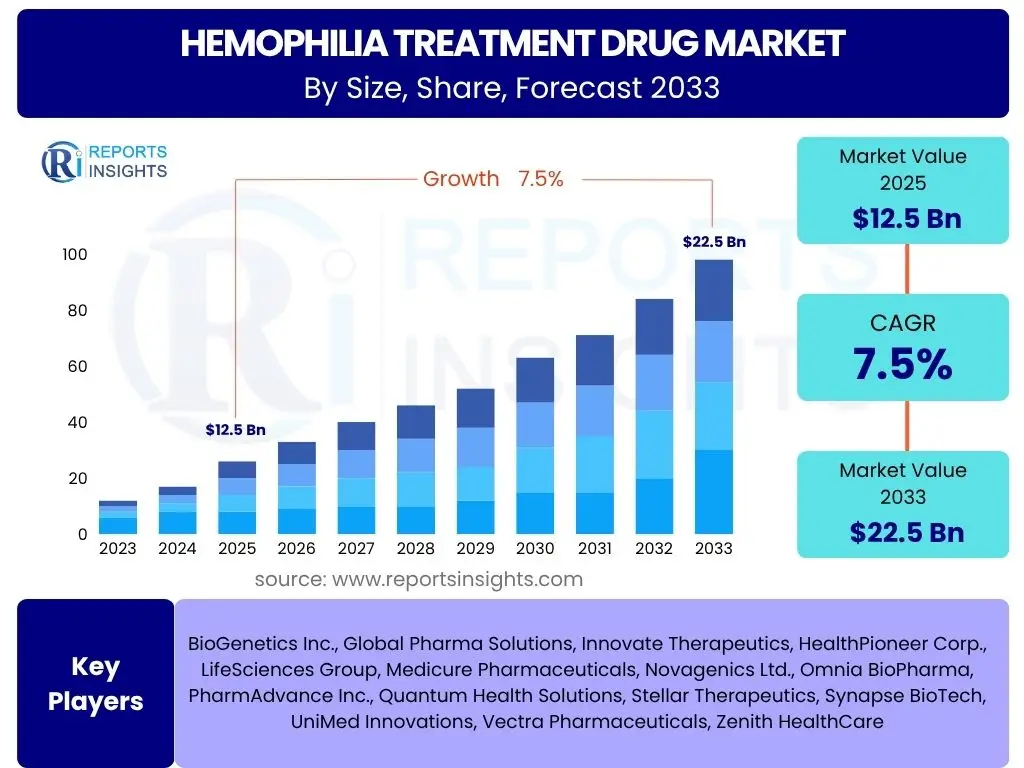
According to Reports Insights Consulting Pvt Ltd, The Hemophilia Treatment Drug Market is projected to grow at a Compound Annual Growth Rate (CAGR) of 7.5% between 2025 and 2033. This growth trajectory reflects significant advancements in therapeutic options and increasing awareness regarding early diagnosis and comprehensive care. The market is propelled by a shift towards prophylaxis, the introduction of novel long-acting therapies, and the emerging potential of gene therapy, which promises transformative treatment outcomes for patients suffering from this chronic bleeding disorder.
The market is estimated at USD 12.5 billion in 2025, a valuation underpinned by the current adoption rates of factor replacement therapies and the initial commercialization of advanced non-factor therapies. This valuation also accounts for the substantial investment in research and development by pharmaceutical companies aimed at addressing unmet needs within the hemophilia community, including patients with inhibitors or those seeking less frequent treatment regimens. The economic burden of hemophilia, coupled with the high cost of existing and pipeline treatments, significantly contributes to the overall market size.
By the end of the forecast period in 2033, the market is projected to reach USD 22.5 billion. This substantial increase is attributed to the anticipated widespread adoption of gene therapies as they gain regulatory approvals and demonstrate long-term efficacy and safety. Furthermore, the expansion of healthcare infrastructure in emerging economies, coupled with improved access to diagnostic tools and treatment, will drive market penetration. The continuous innovation in drug delivery systems and the development of therapies with novel mechanisms of action are expected to sustain robust market growth throughout the forecast period.
Key Hemophilia Treatment Drug Market Trends & Insights
Users frequently inquire about the latest advancements and shifts in hemophilia treatment. A primary area of interest revolves around the transition from conventional factor replacement therapies to longer-acting factor concentrates and, increasingly, to non-factor replacement therapies that offer greater convenience and improved patient outcomes. There is also a significant focus on the revolutionary potential of gene therapy, which is perceived as a curative approach, and how it is reshaping the treatment landscape for hemophilia A and B. Furthermore, inquiries highlight the growing emphasis on personalized medicine, data-driven treatment protocols, and home-based care options, reflecting a broader trend towards patient-centric healthcare solutions.
Another key trend concerns the management of inhibitors, a significant complication in hemophilia treatment. The development of bypass agents and immune tolerance induction (ITI) regimens, alongside the emergence of novel non-factor therapies that are effective in patients with inhibitors, represents a critical area of innovation. The increasing global prevalence of hemophilia, coupled with enhanced diagnostic capabilities, particularly in developing regions, is driving demand for both established and novel treatments. Healthcare systems are also exploring more cost-effective and sustainable treatment models, given the high expenditures associated with lifelong hemophilia management, fostering innovation in drug pricing and access strategies.
- Shift towards longer-acting factor replacement therapies to reduce treatment frequency.
- Emergence and rapid adoption of novel non-factor replacement therapies (e.g., Emicizumab, Fitusiran, Concizumab) offering alternative mechanisms of action and subcutaneous administration.
- Significant advancements and commercialization of gene therapies (e.g., Valoctocogene Roxaparvovec, Etranacogene Dezaparvovec) promising durable factor expression.
- Increasing focus on personalized medicine and precision dosing guided by pharmacokinetics and patient-specific factors.
- Expansion of home-based prophylactic treatment for improved patient convenience and quality of life.
- Growing emphasis on inhibitor management through novel bypass agents and non-factor therapies.
- Integration of digital health solutions and remote monitoring for better disease management.
- Increased market penetration in emerging economies due to improving healthcare infrastructure and awareness.
AI Impact Analysis on Hemophilia Treatment Drug
Common user inquiries regarding AI's impact on the Hemophilia Treatment Drug market center on its potential to accelerate drug discovery, enhance diagnostic accuracy, and personalize treatment regimens. Users are keen to understand how artificial intelligence and machine learning algorithms can analyze vast datasets of patient information, genetic profiles, and drug interactions to identify novel therapeutic targets or predict treatment responses. There is also a strong interest in AI's role in streamlining clinical trials, optimizing patient recruitment, and monitoring real-world evidence to improve therapeutic strategies. The overarching expectation is that AI will lead to more efficient, effective, and tailored hemophilia care, ultimately improving patient outcomes and reducing healthcare burdens.
Furthermore, users frequently ask about the ethical considerations and data privacy challenges associated with integrating AI into sensitive medical areas like hemophilia. Concerns regarding algorithmic bias, data security, and the need for robust regulatory frameworks are often raised. However, the perceived benefits of AI in areas such as identifying at-risk populations for complications like inhibitors, or optimizing prophylactic dosing based on individual bleed patterns, generally outweigh these concerns, provided appropriate safeguards are in place. The potential for AI to support remote consultations and disease management in underserved areas is also a significant point of discussion, contributing to a more equitable distribution of advanced care.
- Accelerated drug discovery and development through AI-driven target identification and molecule design, potentially reducing R&D timelines for new hemophilia therapies.
- Enhanced precision in diagnosis and subtyping of hemophilia, including the early detection of inhibitor development, using AI-powered diagnostic tools.
- Personalized treatment planning and dosage optimization based on individual patient genetic profiles, bleed history, and real-time physiological data analysis.
- Optimization of clinical trial design, patient stratification, and outcome prediction, leading to more efficient and successful development of novel drugs.
- Improved adherence to treatment regimens through AI-powered patient monitoring systems, reminders, and predictive analytics for potential non-adherence.
- Better management of patient data and electronic health records, enabling comprehensive insights for research, treatment efficacy assessment, and epidemiological studies.
- Development of AI-powered decision support systems for clinicians, providing evidence-based recommendations for complex treatment scenarios, especially in inhibitor management.
- Predictive analytics for anticipating bleeding episodes or complications, allowing for proactive intervention and prophylaxis adjustment.
Key Takeaways Hemophilia Treatment Drug Market Size & Forecast
Users commonly seek concise summaries of the market's trajectory and the most impactful factors influencing its growth. A key takeaway is the robust expansion driven by therapeutic innovation, particularly the advent of gene therapy and novel non-factor treatments, which are set to transform hemophilia management from lifelong factor infusions to potentially curative or less frequent dosing regimens. The market's growth is also strongly influenced by increasing global awareness, improved diagnostic capabilities, and the rising prevalence of the disorder, all contributing to a larger patient pool requiring advanced care.
Another significant insight revolves around the substantial investment in research and development, indicating a strong pipeline of innovative drugs addressing unmet medical needs, especially for patients with inhibitors. While high treatment costs and accessibility remain challenges, the long-term outlook for the hemophilia treatment market remains highly positive, with a clear trend towards more convenient, effective, and patient-centric therapies. The market is poised for continued evolution, with a focus on improving quality of life for patients and reducing the overall burden of the disease.
- The Hemophilia Treatment Drug Market is experiencing significant growth, primarily fueled by the introduction of groundbreaking therapies.
- Gene therapy and novel non-factor treatments are set to revolutionize the treatment landscape, potentially offering long-term or curative solutions.
- Increased diagnosis rates and growing awareness globally are expanding the addressable patient population.
- Substantial R&D investments by pharmaceutical companies are ensuring a strong pipeline of innovative drugs.
- Despite high costs, the market demonstrates a clear shift towards more convenient, effective, and personalized treatment options.
- Prophylaxis remains the standard of care, with a growing preference for long-acting and subcutaneous formulations.
- Management of inhibitors remains a critical focus area, driving demand for specialized and novel treatments.
Hemophilia Treatment Drug Market Drivers Analysis
The Hemophilia Treatment Drug Market is primarily driven by a confluence of factors that enhance both the demand for and the availability of advanced therapeutic options. A significant driver is the increasing global prevalence of hemophilia, coupled with improved diagnostic rates, particularly in developing regions, which brings more patients into the treatment paradigm. Furthermore, the persistent and substantial investment in research and development by pharmaceutical companies has led to a prolific pipeline of innovative treatments, including longer-acting factor concentrates, non-factor replacement therapies, and the highly anticipated gene therapies, all of which promise better patient outcomes and reduced treatment burden. These advancements are critical in addressing the unmet medical needs of patients, especially those who develop inhibitors to conventional treatments.
| Drivers | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Increasing Prevalence and Diagnosis of Hemophilia | +1.5% | Global, particularly Asia Pacific, Latin America | 2025-2033 (Long-term) |
| Advancements in Therapeutic Options (Long-acting, Non-Factor, Gene Therapy) | +2.0% | North America, Europe, Japan | 2025-2033 (Long-term) |
| Growing Adoption of Prophylactic Treatment Regimens | +1.0% | Global, particularly Developed Markets | 2025-2033 (Long-term) |
| Favorable Reimbursement Policies and Patient Support Programs | +0.8% | North America, Western Europe | 2025-2033 (Mid to Long-term) |
Hemophilia Treatment Drug Market Restraints Analysis
Despite robust growth prospects, the Hemophilia Treatment Drug Market faces significant restraints that can impede its full potential. The exorbitant cost associated with hemophilia treatment, especially for novel therapies like gene therapy and long-acting factor concentrates, represents a major barrier to access and adoption globally. This high cost often strains healthcare budgets and limits patient access in regions with less developed healthcare infrastructures or stringent reimbursement policies. Furthermore, the development of inhibitors against factor replacement therapies in a significant percentage of patients remains a critical clinical challenge, necessitating more expensive and complex bypass agents or immune tolerance induction regimens, thereby increasing the overall treatment burden and cost.
| Restraints | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| High Cost of Hemophilia Treatment and Novel Therapies | -1.2% | Global, particularly Emerging Markets | 2025-2033 (Long-term) |
| Risk of Inhibitor Development in Patients | -0.8% | Global | 2025-2033 (Long-term) |
| Limited Access to Diagnosis and Treatment in Underserved Regions | -0.7% | Africa, parts of Asia Pacific, Latin America | 2025-2033 (Long-term) |
| Complex Regulatory Approval Pathways for Advanced Therapies | -0.5% | Global | 2025-2030 (Mid-term) |
Hemophilia Treatment Drug Market Opportunities Analysis
The Hemophilia Treatment Drug Market presents several lucrative opportunities driven by scientific breakthroughs and expanding market access. The most significant opportunity lies in the continued development and broader adoption of gene therapies for hemophilia A and B, which promise a potential one-time curative treatment, thereby transforming the long-term management of the disorder. This paradigm shift could unlock new patient segments and redefine treatment economics. Furthermore, the pipeline for novel non-factor therapies with different mechanisms of action, subcutaneous administration, and effectiveness in inhibitor patients offers significant market expansion potential, addressing current treatment limitations and improving patient convenience.
| Opportunities | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Expansion of Gene Therapy Applications and Accessibility | +1.8% | North America, Europe, Asia Pacific (Japan) | 2027-2033 (Long-term) |
| Development and Adoption of Novel Non-Factor Therapies | +1.5% | Global | 2025-2033 (Long-term) |
| Untapped Potential in Emerging Markets with Low Diagnosis Rates | +1.0% | Asia Pacific (China, India), Latin America, MEA | 2025-2033 (Long-term) |
| Strategic Collaborations and Partnerships for R&D and Commercialization | +0.7% | Global | 2025-2033 (Long-term) |
Hemophilia Treatment Drug Market Challenges Impact Analysis
The Hemophilia Treatment Drug Market faces several critical challenges that demand strategic navigation from industry stakeholders. A primary challenge is the high manufacturing complexity and cost associated with advanced biological therapies, particularly gene therapies, which require specialized production facilities and rigorous quality control. This complexity can impact scalability and ultimately affect the affordability and accessibility of these treatments globally. Furthermore, ensuring equitable access to these high-cost therapies in diverse healthcare systems, particularly in developing countries, presents a significant hurdle, often compounded by varying reimbursement policies and health insurance coverage limitations.
| Challenges | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| High Manufacturing Complexity and Cost of Advanced Therapies | -0.9% | Global | 2025-2033 (Long-term) |
| Ensuring Equitable Global Access and Affordability | -0.7% | Emerging Markets | 2025-2033 (Long-term) |
| Patient Adherence to Complex Treatment Regimens | -0.6% | Global | 2025-2033 (Long-term) |
| Long-term Safety and Efficacy Data for Gene Therapies | -0.5% | Global | 2025-2030 (Mid-term) |
Hemophilia Treatment Drug Market - Updated Report Scope
This comprehensive report provides an in-depth analysis of the Hemophilia Treatment Drug market, offering critical insights into its current landscape, historical performance, and future projections. The scope encompasses detailed market sizing, growth drivers, restraints, opportunities, and challenges influencing the industry's trajectory. It further segments the market by type of hemophilia, therapy type, route of administration, and end-user, providing a granular view of various market components. Regional analyses highlight key market dynamics across major geographies, while a competitive landscape section profiles key industry players, their strategies, and recent developments, providing a holistic understanding of the market's competitive intensity and strategic alliances.
| Report Attributes | Report Details |
|---|---|
| Base Year | 2024 |
| Historical Year | 2019 to 2023 |
| Forecast Year | 2025 - 2033 |
| Market Size in 2025 | USD 12.5 billion |
| Market Forecast in 2033 | USD 22.5 billion |
| Growth Rate | 7.5% |
| Number of Pages | 265 |
| Key Trends |
|
| Segments Covered |
|
| Key Companies Covered | BioGenetics Inc., Global Pharma Solutions, Innovate Therapeutics, HealthPioneer Corp., LifeSciences Group, Medicure Pharmaceuticals, Novagenics Ltd., Omnia BioPharma, PharmAdvance Inc., Quantum Health Solutions, Stellar Therapeutics, Synapse BioTech, UniMed Innovations, Vectra Pharmaceuticals, Zenith HealthCare |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Speak to Analyst | Avail customised purchase options to meet your exact research needs. Request For Analyst Or Customization |
Segmentation Analysis
The Hemophilia Treatment Drug market is comprehensively segmented to provide a detailed understanding of its diverse components, allowing for targeted analysis of growth drivers and market dynamics within specific categories. These segmentations are crucial for identifying emerging opportunities, assessing competitive landscapes, and formulating effective market strategies across various patient populations and treatment modalities. The primary segments include categorization by the specific type of hemophilia, the broad therapeutic approaches employed, the route through which these treatments are administered, and the settings where patients receive care.
- By Hemophilia Type: This segment differentiates therapies based on the specific factor deficiency.
- Hemophilia A: Dominant segment due to higher prevalence, focusing on Factor VIII replacement and related novel therapies.
- Hemophilia B: Addresses Factor IX deficiency, with significant advancements in long-acting and gene therapies.
- Others: Includes rare forms like Hemophilia C and acquired hemophilia, requiring specialized treatment approaches.
- By Therapy Type: This segment examines the mechanism of action of the treatments.
- Factor Replacement Therapy: The traditional cornerstone of treatment, involving the infusion of deficient clotting factors.
- Factor VIII
- Factor IX
- Non-Factor Replacement Therapy: Represents a growing and innovative segment, offering alternative mechanisms to promote hemostasis.
- Bypassing Agents: Used for patients with inhibitors.
- Gene Therapy: A transformative approach aiming for a functional cure.
- Other Novel Therapies: Includes RNAi therapies, bispecific antibodies, and other emerging non-factor approaches.
- By Route of Administration: This segment highlights the preferred method of drug delivery, influencing patient convenience and adherence.
- Intravenous: Traditional method for factor replacement therapies.
- Subcutaneous: Gaining prominence due to convenience, especially for novel non-factor therapies.
- By End-User: This segment identifies the primary settings where hemophilia treatments are administered.
- Hospitals: Major centers for initial diagnosis, severe bleeds, and complex treatment initiation.
- Homecare Settings: Increasing preference for routine prophylactic infusions due to convenience and improved patient quality of life.
- Specialty Clinics: Dedicated centers offering comprehensive care, diagnostics, and management.
Regional Highlights
- North America: Dominates the Hemophilia Treatment Drug market due to high healthcare expenditure, early adoption of advanced therapies, robust research and development activities, and favorable reimbursement policies. The United States accounts for the largest share, driven by a large patient pool and access to cutting-edge treatments.
- Europe: A significant market driven by increasing awareness, well-established healthcare infrastructure, and the presence of key pharmaceutical companies. Countries like Germany, the UK, France, Italy, and Spain are leading the adoption of new therapies and have strong patient advocacy groups.
- Asia Pacific (APAC): Emerging as the fastest-growing market due to improving healthcare infrastructure, rising disposable incomes, increasing diagnosis rates, and a large patient population in countries like China, India, and Japan. Government initiatives to improve access to treatment are further boosting market growth.
- Latin America: Experiences moderate growth, with increasing efforts to improve hemophilia care and diagnosis. Brazil and Mexico are key markets, benefiting from growing healthcare investments and patient support programs, though access to high-cost therapies remains a challenge.
- Middle East and Africa (MEA): Represents a nascent market with significant untapped potential. Growth is driven by increasing awareness, improving diagnostic capabilities, and governmental focus on healthcare infrastructure development, particularly in GCC countries and South Africa. However, economic disparities and limited access to specialized care remain prevalent.
Top Key Players
The market research report includes a detailed profile of leading stakeholders in the Hemophilia Treatment Drug Market.- BioGenetics Inc.
- Global Pharma Solutions
- Innovate Therapeutics
- HealthPioneer Corp.
- LifeSciences Group
- Medicure Pharmaceuticals
- Novagenics Ltd.
- Omnia BioPharma
- PharmAdvance Inc.
- Quantum Health Solutions
- Stellar Therapeutics
- Synapse BioTech
- UniMed Innovations
- Vectra Pharmaceuticals
- Zenith HealthCare
- Apex Therapeutics
- CurePath Bio
- GeneForm Biotech
- Regenesis Pharma
- Vitalis MedTech
Frequently Asked Questions
Analyze common user questions about the Hemophilia Treatment Drug market and generate a concise list of summarized FAQs reflecting key topics and concerns.What is hemophilia and how is it treated?
Hemophilia is a rare genetic bleeding disorder where the blood does not clot properly due to a deficiency in specific clotting factors. Treatment primarily involves factor replacement therapy to provide the missing clotting factor, often administered prophylactically to prevent bleeding episodes. Newer non-factor therapies and gene therapies are also emerging as significant treatment options.
What are the latest innovations in hemophilia treatment?
The latest innovations include longer-acting factor concentrates that reduce infusion frequency, novel non-factor therapies (such as bispecific antibodies and RNA interference agents) offering subcutaneous administration and different mechanisms of action, and gene therapies that aim to provide a durable, potentially curative solution by enabling the body to produce its own clotting factor.
How large is the global hemophilia treatment drug market and what is its growth forecast?
The global Hemophilia Treatment Drug market is estimated at USD 12.5 billion in 2025 and is projected to reach USD 22.5 billion by 2033, growing at a Compound Annual Growth Rate (CAGR) of 7.5%. This growth is driven by therapeutic advancements, increasing diagnosis rates, and expanding access to care.
What challenges does the hemophilia treatment market face?
Key challenges include the high cost of existing and novel therapies, the development of inhibitors in some patients which complicates treatment, limited access to diagnosis and specialized care in developing regions, and the complex manufacturing and regulatory pathways for advanced biological drugs, particularly gene therapies.
How is gene therapy impacting the hemophilia treatment landscape?
Gene therapy is revolutionizing hemophilia treatment by offering the potential for a one-time, long-lasting solution, reducing or eliminating the need for regular factor infusions. While still in early stages of commercialization, it promises to significantly improve patient quality of life and reduce the long-term burden of the disease by enabling endogenous factor production.

