Entyvio Drug Market
Entyvio Drug Market Size, Scope, Growth, Trends and By Segmentation Types, Applications, Regional Analysis and Industry Forecast (2025-2033)
Report ID : RI_701298 | Last Updated : July 29, 2025 |
Format : ![]()
![]()
![]()
![]()
Entyvio Drug Market Size
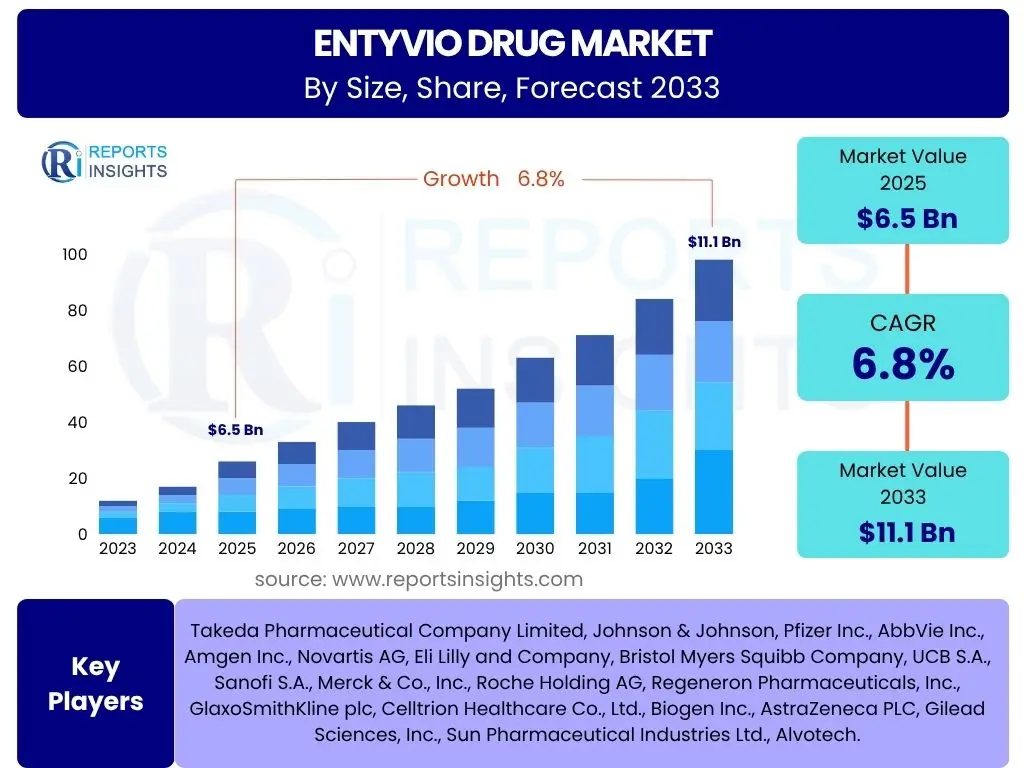
According to Reports Insights Consulting Pvt Ltd, The Entyvio Drug Market is projected to grow at a Compound Annual Growth Rate (CAGR) of 6.8% between 2025 and 2033. The market is estimated at USD 6.5 billion in 2025 and is projected to reach USD 11.1 billion by the end of the forecast period in 2033. This growth trajectory is significantly influenced by the increasing global prevalence of inflammatory bowel diseases (IBD), including Crohn's disease and ulcerative colitis, for which Entyvio (vedolizumab) offers a targeted therapeutic option. The expanding adoption of biologic therapies due to their improved efficacy and safety profiles compared to conventional treatments also underpins this robust market expansion.
The market's expansion is further propelled by advancements in diagnostic methodologies leading to earlier and more accurate diagnosis of IBD, thereby increasing the patient pool eligible for biologic treatments. Enhanced patient access to advanced healthcare facilities and greater healthcare expenditure in developing regions are contributing factors. Moreover, the increasing awareness among both healthcare professionals and patients regarding the long-term benefits of biologic interventions for chronic gastrointestinal conditions is driving prescription rates and market demand. Strategic initiatives such as patient support programs and educational campaigns by pharmaceutical companies also play a crucial role in fostering market growth and ensuring sustained patient adherence to treatment regimens.
Key Entyvio Drug Market Trends & Insights
Users frequently inquire about the evolving landscape of the Entyvio drug market, focusing on factors shaping its future and competitive dynamics. Key trends indicate a significant shift towards personalized medicine approaches within IBD treatment, driven by a deeper understanding of disease heterogeneity and individual patient responses to biologics. There is also a notable emphasis on optimizing drug delivery methods, with a growing interest in subcutaneous formulations to enhance patient convenience and adherence. Furthermore, the market is experiencing intensified competition from emerging biologics and biosimilars, prompting continuous innovation in treatment strategies and patient management.
Another prominent trend involves the integration of real-world evidence (RWE) into clinical practice and drug development. RWE helps in demonstrating the long-term effectiveness and safety of Entyvio in diverse patient populations, thereby strengthening its market position and informing treatment guidelines. The focus on early intervention with biologics, particularly in moderate-to-severe IBD cases, is also gaining traction, aiming to prevent disease progression and improve patient outcomes. These trends collectively underscore a dynamic market environment characterized by continuous innovation, patient-centric solutions, and a strategic response to evolving clinical needs and competitive pressures.
- Growing adoption of personalized medicine in IBD treatment based on patient biomarkers and disease phenotype.
- Development and increased availability of subcutaneous formulations for enhanced patient convenience and adherence.
- Escalating competition from novel biologics and the anticipated entry of biosimilars impacting market share.
- Increasing emphasis on real-world evidence (RWE) to validate long-term efficacy and safety of Entyvio.
- Shift towards early and aggressive biologic therapy in moderate-to-severe inflammatory bowel disease.
- Integration of digital health solutions and patient support programs to improve treatment outcomes and adherence.
AI Impact Analysis on Entyvio Drug
Common user questions regarding AI's influence on the Entyvio drug market revolve around its potential to revolutionize patient diagnosis, treatment pathways, and drug development processes. Users are keen to understand how artificial intelligence can optimize drug selection, predict treatment response, and enhance patient monitoring for chronic conditions like IBD. The underlying expectation is that AI will streamline clinical workflows, reduce healthcare costs, and ultimately lead to more effective and personalized patient care, thereby expanding the applicability and efficacy of treatments like Entyvio.
AI's impact extends beyond clinical applications, influencing market dynamics through improved R&D efficiency, accelerated clinical trials, and advanced market forecasting. It holds the promise of identifying new patient cohorts, optimizing pricing strategies, and refining distribution networks for specialty drugs. Furthermore, AI-driven insights from large datasets, including electronic health records and real-world data, can inform post-market surveillance and identify opportunities for label expansion or new therapeutic combinations, thereby enhancing the overall value proposition of Entyvio and similar biologics in a competitive market.
- Accelerated drug discovery and development processes for new IBD therapies.
- Enhanced precision in patient stratification and prediction of Entyvio treatment response.
- Optimization of clinical trial design and patient recruitment for IBD studies.
- Improved real-time patient monitoring and disease management for IBD patients on Entyvio.
- Development of AI-powered diagnostic tools for earlier and more accurate IBD detection.
- Streamlined supply chain and inventory management for specialty pharmaceuticals.
- Personalized dosage and treatment regimen recommendations based on individual patient data.
Key Takeaways Entyvio Drug Market Size & Forecast
Users frequently seek concise summaries of the Entyvio drug market's future outlook and key growth drivers. The primary takeaway is the significant and sustained growth projected for the market, driven by the increasing global burden of inflammatory bowel diseases and the growing preference for advanced biologic therapies. The forecast indicates a robust expansion, reinforcing Entyvio's established position as a leading treatment option for ulcerative colitis and Crohn's disease, with its market share bolstered by strong clinical efficacy and a favorable safety profile.
Another crucial insight is the dynamic interplay of market opportunities, such as expanding indications and geographic reach, tempered by inherent challenges like the emergence of biosimilars and the high cost of treatment. Strategic responses to these factors, including innovative patient support programs and potential for new formulations, will be critical for sustained growth. The market's future trajectory will also be shaped by the pace of medical advancements, regulatory changes, and the ability of manufacturers to address unmet patient needs while navigating a competitive landscape.
- The Entyvio drug market is poised for significant growth, reaching USD 11.1 billion by 2033, driven by IBD prevalence.
- Strong clinical efficacy and safety profile of Entyvio underpin its sustained market dominance.
- Increasing adoption of biologic therapies for chronic gastrointestinal conditions is a key growth propeller.
- Opportunities exist in expanding therapeutic indications and geographic penetration, particularly in emerging markets.
- Challenges include intense competition from biosimilars and high treatment costs, necessitating strategic market responses.
- Personalized medicine and advanced diagnostic tools will increasingly shape future treatment paradigms.
Entyvio Drug Market Drivers Analysis
The Entyvio drug market is propelled by a confluence of factors, primarily the escalating global incidence and prevalence of inflammatory bowel diseases (IBD), including Crohn's disease and ulcerative colitis. As these chronic conditions become more widespread, the demand for effective and targeted therapies like Entyvio naturally increases. Awareness campaigns, improved diagnostic capabilities, and greater access to healthcare services in various regions also contribute significantly to identifying more patients eligible for advanced biologic treatments. The shift towards biologic therapies, known for their superior efficacy and improved safety profiles compared to conventional immunosuppressants, further accelerates market growth.
Additionally, favorable reimbursement policies in developed markets and increasing healthcare expenditure in emerging economies are making biologic treatments more accessible to a broader patient population. Ongoing research and development efforts continue to reinforce the long-term benefits and effectiveness of existing biologics while exploring new indications or combination therapies. The aging global population, which often presents with a higher incidence of chronic diseases, also adds to the patient pool requiring long-term management with drugs like Entyvio.
| Drivers | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Rising Incidence of Inflammatory Bowel Diseases (IBD) | +1.5% | Global, particularly North America, Europe, Asia Pacific | Short to Long-term (2025-2033) |
| Increasing Adoption of Biologic Therapies | +1.2% | Global, especially Developed Markets | Short to Mid-term (2025-2029) |
| Advancements in Diagnostic Technologies | +0.8% | North America, Europe | Mid-term (2027-2033) |
| Favorable Reimbursement Policies & Healthcare Expenditure | +0.7% | North America, Western Europe, Japan | Ongoing (2025-2033) |
| Growing Awareness of IBD and Treatment Options | +0.6% | Emerging Economies (APAC, Latin America) | Mid to Long-term (2027-2033) |
Entyvio Drug Market Restraints Analysis
Despite robust growth, the Entyvio drug market faces several significant restraints that could temper its expansion. A primary limiting factor is the high cost associated with biologic therapies. These treatments often represent a substantial financial burden for healthcare systems and individual patients, especially in regions with limited insurance coverage or lower per capita income. This high cost can lead to restricted access, delayed treatment initiation, or even non-adherence, thereby limiting the overall patient pool receiving Entyvio.
Another substantial restraint is the increasing market saturation and intense competition from existing and pipeline biologics, as well as the anticipated entry of biosimilars. As patents expire, the emergence of lower-cost biosimilar alternatives will exert downward pressure on pricing and market share, potentially eroding the revenue of branded drugs like Entyvio. Additionally, stringent regulatory approval processes for new indications or formulations, coupled with potential side effects or a lack of response in certain patient populations, pose ongoing challenges for market expansion. The long-term safety profile and the need for continuous monitoring also add complexity and potential costs to treatment, which can be a barrier for some patients or healthcare providers.
| Restraints | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| High Cost of Biologic Therapies | -1.3% | Global, particularly developing and cost-sensitive markets | Short to Long-term (2025-2033) |
| Emergence of Biosimilars and Generic Competition | -1.0% | North America, Europe | Mid to Long-term (Post-2028) |
| Stringent Regulatory Approval Processes | -0.6% | Global | Ongoing (2025-2033) |
| Risk of Adverse Effects and Immunogenicity | -0.4% | Global | Ongoing (2025-2033) |
| Lack of Response in Certain Patient Populations | -0.3% | Global | Ongoing (2025-2033) |
Entyvio Drug Market Opportunities Analysis
The Entyvio drug market presents significant opportunities for sustained growth and expansion. One key area is the untapped patient population, especially in emerging markets, where diagnosis rates for IBD are lower, and access to advanced therapies is limited. As healthcare infrastructure improves and economic conditions strengthen in these regions, there will be a substantial increase in demand for effective treatments like Entyvio. Furthermore, the exploration of new therapeutic indications beyond ulcerative colitis and Crohn's disease, or in specific sub-populations of IBD patients, could unlock new revenue streams and expand the drug's utility.
Another major opportunity lies in the development of more convenient drug delivery methods, such as subcutaneous formulations, which can significantly enhance patient adherence and quality of life. The integration of digital health solutions, including remote patient monitoring, telemedicine, and personalized treatment platforms, offers avenues to improve patient management and optimize treatment outcomes. Additionally, strategic collaborations and partnerships with other pharmaceutical companies, research institutions, and healthcare providers can facilitate market penetration, accelerate R&D, and overcome regulatory hurdles, further solidifying Entyvio's market position and reach.
| Opportunities | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Expansion into Emerging Markets (Untapped Patient Pool) | +1.1% | Asia Pacific, Latin America, Middle East & Africa | Mid to Long-term (2027-2033) |
| Development of Subcutaneous Formulations for Improved Convenience | +0.9% | Global | Mid-term (2027-2030) |
| Exploration of New Therapeutic Indications or Sub-populations | +0.8% | Global | Long-term (2030-2033) |
| Integration of Digital Health and Personalized Medicine Solutions | +0.7% | North America, Europe | Mid to Long-term (2027-2033) |
| Strategic Partnerships and Collaborations | +0.5% | Global | Ongoing (2025-2033) |
Entyvio Drug Market Challenges Impact Analysis
The Entyvio drug market faces several persistent challenges that could impede its growth trajectory. One significant hurdle is the escalating competition from a growing pipeline of novel biologics and small molecule drugs targeting IBD. This intense rivalry necessitates continuous innovation and differentiation to maintain market share. Additionally, the increasing pressure from payers and healthcare systems to control costs, particularly for high-cost biologics, leads to tougher negotiation environments, restricted formularies, and an emphasis on value-based care, potentially limiting widespread adoption.
Another challenge stems from patient access and adherence issues. Despite the drug's efficacy, factors such as the need for long-term administration, potential for non-response or loss of response over time, and the complexity of managing a chronic disease can lead to patient dropouts or suboptimal outcomes. Global supply chain vulnerabilities, manufacturing complexities associated with biologic drugs, and the evolving regulatory landscape further add to the operational challenges for manufacturers. Moreover, the lack of definitive biomarkers to predict individual patient response to Entyvio can lead to trial-and-error prescribing, delaying effective treatment and increasing healthcare costs.
| Challenges | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Intense Competition from Pipeline and Approved Therapies | -0.9% | Global | Short to Mid-term (2025-2029) |
| Pressure on Pricing and Reimbursement Policies | -0.8% | North America, Europe | Ongoing (2025-2033) |
| Patient Non-adherence and Loss of Response Over Time | -0.5% | Global | Ongoing (2025-2033) |
| Manufacturing Complexities and Supply Chain Vulnerabilities | -0.4% | Global | Ongoing (2025-2033) |
| Lack of Predictive Biomarkers for Treatment Response | -0.3% | Global | Long-term (2030-2033) |
Entyvio Drug Market - Updated Report Scope
This updated report provides an in-depth analysis of the Entyvio drug market, covering historical performance, current market dynamics, and future projections. It meticulously examines market size, growth drivers, restraints, opportunities, and challenges that shape the industry landscape. The report also offers a comprehensive segmentation analysis by indication, route of administration, distribution channel, and end-user, complemented by a detailed regional outlook. Furthermore, it incorporates an AI impact analysis and an exhaustive profiling of key market players, offering strategic insights for stakeholders to navigate the evolving market environment and capitalize on emerging trends.
| Report Attributes | Report Details |
|---|---|
| Base Year | 2024 |
| Historical Year | 2019 to 2023 |
| Forecast Year | 2025 - 2033 |
| Market Size in 2025 | USD 6.5 Billion |
| Market Forecast in 2033 | USD 11.1 Billion |
| Growth Rate | 6.8% CAGR |
| Number of Pages | 245 |
| Key Trends |
|
| Segments Covered |
|
| Key Companies Covered | Takeda Pharmaceutical Company Limited, Johnson & Johnson, Pfizer Inc., AbbVie Inc., Amgen Inc., Novartis AG, Eli Lilly and Company, Bristol Myers Squibb Company, UCB S.A., Sanofi S.A., Merck & Co., Inc., Roche Holding AG, Regeneron Pharmaceuticals, Inc., GlaxoSmithKline plc, Celltrion Healthcare Co., Ltd., Biogen Inc., AstraZeneca PLC, Gilead Sciences, Inc., Sun Pharmaceutical Industries Ltd., Alvotech. |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Speak to Analyst | Avail customised purchase options to meet your exact research needs. Request For Analyst Or Customization |
Segmentation Analysis
The Entyvio drug market is meticulously segmented to provide a granular understanding of its diverse components and drivers. This segmentation allows for precise analysis of patient demographics, treatment preferences, and market penetration across various applications and distribution channels. The primary segmentations include differentiation by the specific inflammatory bowel disease indication, the route of administration for the drug, the channels through which the drug is distributed to patients, and the ultimate end-users of the therapy. Each segment exhibits unique growth patterns and market dynamics, contributing distinctively to the overall market valuation.
Understanding these segments is crucial for stakeholders to identify high-growth areas, develop targeted marketing strategies, and optimize resource allocation. For instance, the market for Entyvio in Crohn's disease might exhibit different growth rates and competitive landscapes compared to ulcerative colitis, necessitating distinct commercial approaches. Similarly, the shift from intravenous to subcutaneous administration impacts patient convenience and healthcare resource utilization, influencing market adoption and preferences. Analyzing these granular segments provides actionable insights into the complexities of the Entyvio drug market and its future trajectory.
- By Indication:
- Ulcerative Colitis: This segment accounts for a significant share due to the established efficacy of Entyvio in inducing and maintaining remission in UC patients.
- Crohn's Disease: Growing prevalence and increasing therapeutic options contribute to the expansion of this segment.
- Others: Includes off-label uses or investigational applications for other inflammatory conditions where Entyvio might show efficacy.
- By Route of Administration:
- Intravenous: The traditional and primary route of administration for Entyvio, typically administered in a clinical setting.
- Subcutaneous: An emerging and increasingly preferred route offering greater patient convenience for at-home administration, driving future growth.
- By Distribution Channel:
- Hospital Pharmacies: Major channel for initial administration and ongoing supply due to the nature of biologic therapies.
- Retail Pharmacies: Increasing role, especially with the introduction of subcutaneous formulations, for direct patient access.
- Online Pharmacies: A growing segment driven by convenience and digital healthcare trends, though less prominent for initial biologic prescriptions.
- By End-User:
- Hospitals: Primary end-user segment for intravenous administration and complex patient management.
- Clinics: Including gastroenterology clinics and infusion centers, offering specialized care and administration services.
- Ambulatory Surgical Centers: Providing out-patient services for drug administration.
- Others: Includes research institutions or home-care settings.
Regional Highlights
- North America: This region dominates the Entyvio drug market, largely driven by the high prevalence of IBD, advanced healthcare infrastructure, high per capita healthcare spending, and favorable reimbursement policies. The presence of major pharmaceutical companies and robust R&D activities also contributes significantly to market leadership. The U.S. remains the largest market within this region, characterized by rapid adoption of novel therapies and strong patient awareness.
- Europe: The second-largest market for Entyvio, characterized by a well-established healthcare system and increasing awareness of IBD. Countries like Germany, France, and the UK are key contributors, benefiting from strong government support for chronic disease management and a growing emphasis on biologic therapies. However, price controls and healthcare austerity measures in some nations can influence market access and growth.
- Asia Pacific (APAC): Expected to witness the highest growth rate during the forecast period. This surge is attributed to improving healthcare infrastructure, rising disposable incomes, increasing awareness about IBD, and the large patient pool in populous countries like China and India. The expanding geriatric population and increasing prevalence of lifestyle-related diseases also contribute to the rising demand for advanced IBD treatments.
- Latin America: This region is an emerging market with significant growth potential, driven by improving economic conditions, expanding healthcare access, and a growing recognition of IBD. Countries like Brazil and Mexico are leading the adoption of advanced therapies, though challenges related to affordability and healthcare infrastructure remain.
- Middle East and Africa (MEA): Represents a nascent but growing market for Entyvio. Growth is spurred by increasing investments in healthcare infrastructure, rising awareness of chronic diseases, and efforts to improve patient access to specialty drugs. However, political instability and socio-economic disparities in certain sub-regions can hinder uniform market development.
Top Key Players
The market research report includes a detailed profile of leading stakeholders in the Entyvio Drug Market.- Takeda Pharmaceutical Company Limited
- Johnson & Johnson
- Pfizer Inc.
- AbbVie Inc.
- Amgen Inc.
- Novartis AG
- Eli Lilly and Company
- Bristol Myers Squibb Company
- UCB S.A.
- Sanofi S.A.
- Merck & Co., Inc.
- Roche Holding AG
- Regeneron Pharmaceuticals, Inc.
- GlaxoSmithKline plc
- Celltrion Healthcare Co., Ltd.
- Biogen Inc.
- AstraZeneca PLC
- Gilead Sciences, Inc.
- Sun Pharmaceutical Industries Ltd.
- Alvotech
Frequently Asked Questions
What is Entyvio used for?
Entyvio (vedolizumab) is a biologic medication primarily used to treat adults with moderately to severely active ulcerative colitis (UC) and Crohn's disease (CD). It works by selectively targeting and blocking the migration of certain white blood cells into the gut, reducing inflammation without broadly suppressing the immune system.
How is Entyvio administered?
Entyvio is primarily administered as an intravenous (IV) infusion, typically given by a healthcare professional in a clinic or hospital setting. A subcutaneous (SC) formulation is also available in some regions, allowing for self-administration at home after initial IV induction, offering greater patient convenience.
What are the typical side effects of Entyvio?
Common side effects of Entyvio include headache, nausea, joint pain, common cold symptoms (nasopharyngitis), and upper respiratory tract infections. Serious but less common side effects can include infusion-related reactions, infections, and liver problems. Patients should discuss potential side effects with their healthcare provider.
How does Entyvio compare to other IBD biologics?
Entyvio is gut-selective, meaning it primarily affects the gut's immune response, potentially leading to a lower risk of systemic side effects compared to non-gut-selective biologics. It is often considered for patients who have not responded adequately to or cannot tolerate other conventional IBD therapies or TNF blockers. Its efficacy profile in inducing and maintaining remission is well-established.
What is the future outlook for the Entyvio drug market?
The Entyvio drug market is projected for significant growth, driven by the increasing global prevalence of IBD and the rising adoption of biologic therapies. Future growth will also be influenced by the development of more convenient formulations (e.g., subcutaneous), expansion into new geographic markets, and ongoing research into its efficacy in specific patient populations, despite the challenges posed by biosimilar competition.

