Clotting Factor Market
Clotting Factor Market Size, Scope, Growth, Trends and By Segmentation Types, Applications, Regional Analysis and Industry Forecast (2025-2033)
Report ID : RI_703345 | Last Updated : August 01, 2025 |
Format : ![]()
![]()
![]()
![]()
Clotting Factor Market Size
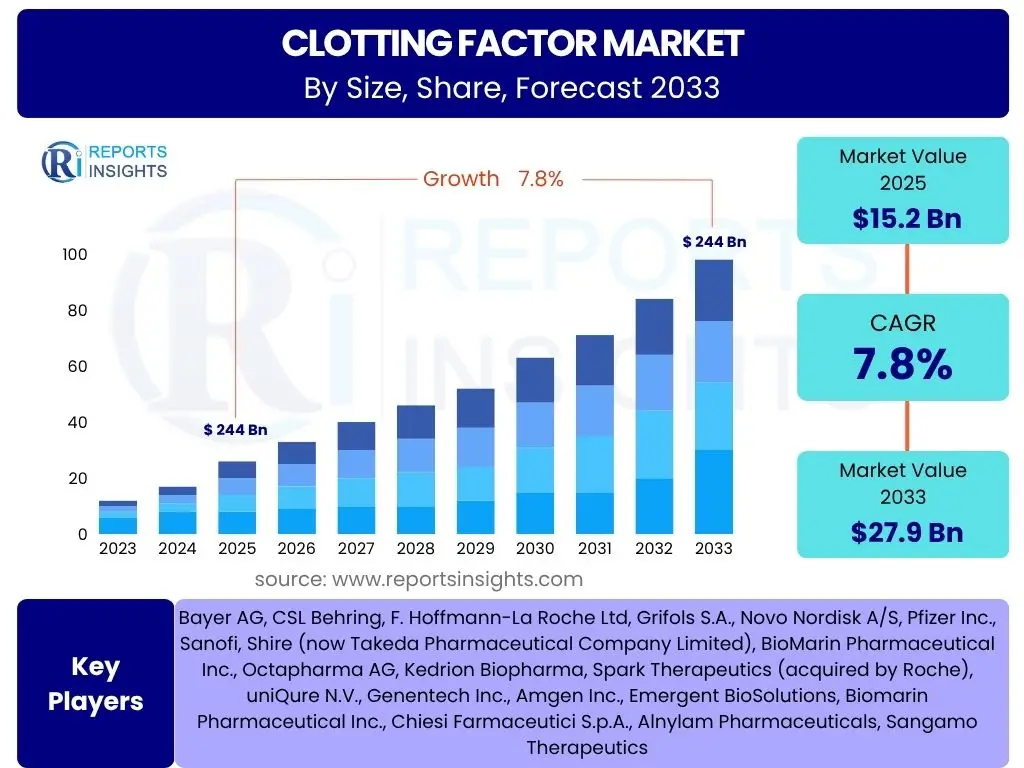
According to Reports Insights Consulting Pvt Ltd, The Clotting Factor Market is projected to grow at a Compound Annual Growth Rate (CAGR) of 7.8% between 2025 and 2033. The market is estimated at USD 15.2 billion in 2025 and is projected to reach USD 27.9 billion by the end of the forecast period in 2033.
Key Clotting Factor Market Trends & Insights
The Clotting Factor market is witnessing a transformative period driven by significant advancements in therapeutic modalities and an increasing global burden of bleeding disorders. Key trends revolve around the continuous innovation in recombinant factor technology, leading to the development of extended half-life (EHL) products that significantly reduce the frequency of infusions, thereby improving patient adherence and quality of life. There is also a pronounced shift towards personalized medicine, where treatment regimens are tailored to individual patient needs, genetics, and specific disease characteristics, optimizing therapeutic outcomes. Furthermore, the market is experiencing a surge in gene therapy research and clinical trials, holding the promise of a potential one-time cure for certain bleeding disorders, which, if successful, could fundamentally reshape the treatment landscape. The focus on orphan drug designations and therapies for rare bleeding disorders beyond hemophilia A and B is also expanding, addressing previously unmet medical needs and broadening the market scope. Finally, the growing awareness and improved diagnostic capabilities globally, particularly in emerging economies, are contributing to an increased patient pool seeking treatment.
- Development and adoption of Extended Half-Life (EHL) clotting factors enhancing convenience and patient adherence.
- Rising prevalence of gene therapy clinical trials and approvals for hemophilia, offering curative potential.
- Increasing focus on personalized treatment approaches and precision medicine for bleeding disorders.
- Growing investment in research and development for novel non-factor therapies and bypassing agents.
- Expansion of diagnostic capabilities and awareness campaigns, leading to improved identification of patients.
- Strategic collaborations and partnerships among pharmaceutical companies and research institutions to accelerate innovation.
AI Impact Analysis on Clotting Factor
The integration of Artificial Intelligence (AI) and Machine Learning (ML) is poised to revolutionize various aspects of the Clotting Factor market, from drug discovery and development to patient management and manufacturing. AI algorithms are increasingly being utilized to analyze vast datasets, including genomic, proteomic, and clinical trial data, to identify novel drug targets, predict patient responses to therapies, and optimize drug formulations. This capability can accelerate the discovery of more effective and safer clotting factor analogues or alternative therapies. In clinical development, AI can enhance trial design, patient recruitment, and data analysis, potentially shortening development timelines and reducing costs for new treatments. For patient care, AI-powered predictive analytics can help monitor disease progression, anticipate bleed events, and optimize prophylactic treatment regimens, leading to more proactive and personalized patient management strategies. Furthermore, AI and automation are expected to streamline manufacturing processes for biologics, improving efficiency, reducing variability, and ensuring higher quality and consistent supply of clotting factors.
- Accelerated drug discovery and target identification through AI-driven data analysis.
- Enhanced clinical trial design, patient selection, and outcome prediction using machine learning.
- Optimization of manufacturing processes for clotting factors, leading to improved efficiency and quality control.
- Development of AI-powered diagnostic tools for earlier and more accurate identification of bleeding disorders.
- Personalized treatment recommendations and patient monitoring through AI-driven predictive analytics.
- Improved supply chain management and inventory optimization for clotting factor products.
Key Takeaways Clotting Factor Market Size & Forecast
The Clotting Factor market is on a robust growth trajectory, primarily fueled by a rising global incidence of bleeding disorders and continuous advancements in therapeutic solutions. The significant projected increase in market size underscores the critical demand for effective and patient-friendly treatments, particularly for conditions like hemophilia. The shift towards innovative therapies such as extended half-life factors and gene therapy signifies a profound evolution in patient care, promising improved quality of life and potentially curative options. This growth is also underpinned by expanding access to diagnosis and treatment in emerging economies, alongside a proactive regulatory environment that supports orphan drug development. Stakeholders should recognize the imperative for continued investment in research and development, focusing on gene therapies and non-factor alternatives, as these innovations are set to be primary growth drivers. Furthermore, strategic collaborations and efforts to address treatment affordability and accessibility will be crucial for sustainable market expansion and reaching underserved populations.
- The market exhibits strong growth potential, driven by unmet medical needs in bleeding disorders.
- Technological advancements, especially in extended half-life factors and gene therapy, are pivotal to market expansion.
- Increasing awareness, improved diagnosis, and expanded treatment access contribute significantly to market growth.
- High treatment costs and the need for greater patient access remain critical considerations for market development.
- The market is shifting towards more patient-centric and personalized treatment approaches.
- Emerging markets present substantial opportunities for growth due to increasing healthcare infrastructure and awareness.
Clotting Factor Market Drivers Analysis
The escalating global prevalence of bleeding disorders, particularly hemophilia, stands as a primary driver for the Clotting Factor market. Conditions like hemophilia A and B, von Willebrand disease, and rare factor deficiencies necessitate lifelong replacement therapy, creating a sustained demand for clotting factor products. As diagnostic capabilities improve, more individuals are accurately identified and brought into the treatment paradigm, further expanding the patient pool requiring these critical therapies. This increasing patient base, coupled with better understanding and management of these chronic conditions, ensures a consistent and growing market for both established and novel clotting factor products.
Technological advancements in the development of recombinant clotting factors represent another significant market driver. The shift from plasma-derived factors to recombinant DNA technology has greatly enhanced product safety by eliminating the risk of pathogen transmission. More recently, the development of extended half-life (EHL) clotting factors has revolutionized treatment by reducing the frequency of intravenous infusions, thereby improving patient adherence, convenience, and overall quality of life, particularly for pediatric patients and those requiring lifelong prophylaxis. These innovations not only address existing patient needs but also attract new patients who might have been hesitant due to the burden of frequent infusions.
Favorable government policies, reimbursement scenarios, and increasing healthcare expenditure, particularly in developed regions, also play a crucial role. Many countries recognize bleeding disorders as chronic, life-threatening conditions and provide robust support for treatment, including significant reimbursement for expensive clotting factor therapies. Orphan drug designations for rare bleeding disorders further incentivize pharmaceutical companies to invest in research and development, ensuring a pipeline of new and improved treatments. The global rise in healthcare infrastructure and improved access to specialized care, even in developing nations, contributes to a broader adoption of clotting factor therapies, further propelling market growth.
| Drivers | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Increasing Global Prevalence of Bleeding Disorders | +1.5% to +2.0% | Global, particularly North America, Europe, Asia Pacific | 2025-2033 (Long-term) |
| Advancements in Recombinant and Extended Half-Life (EHL) Factors | +1.0% to +1.5% | Global, especially Developed Markets | 2025-2030 (Mid-term) |
| Growing Awareness and Improved Diagnostic Capabilities | +0.8% to +1.2% | Emerging Economies (APAC, LATAM, MEA) | 2025-2033 (Long-term) |
| Favorable Reimbursement Policies and Government Support | +0.5% to +0.8% | North America, Western Europe | 2025-2033 (Long-term) |
Clotting Factor Market Restraints Analysis
The exceedingly high cost associated with clotting factor therapies represents a significant restraint on market growth, particularly in regions with limited healthcare budgets or inadequate insurance coverage. Lifelong prophylactic treatment, especially with advanced recombinant and extended half-life factors, can incur annual costs ranging from hundreds of thousands to over a million USD per patient. This financial burden poses a substantial challenge for healthcare systems and can limit patient access, especially in developing countries where a large portion of the global hemophilia population resides. The affordability crisis necessitates innovative pricing models or greater governmental subsidies to ensure equitable access to these life-saving treatments.
The risk of inhibitor development in patients receiving clotting factor replacement therapy is another major concern acting as a market restraint. Inhibitors are antibodies that neutralize the infused clotting factor, rendering the treatment ineffective and leading to severe bleeding episodes. Managing patients with inhibitors is complex, requiring alternative, often more expensive, bypassing agents, which adds to the overall treatment cost and complexity. While efforts are ongoing to reduce inhibitor incidence, its unpredictable nature and significant clinical implications continue to pose a challenge to widespread and uninterrupted therapy, impacting patient outcomes and market predictability.
Stringent regulatory approval processes and the complexities involved in manufacturing highly specialized biologic products also serve as restraints. Clotting factors are complex biologics, requiring rigorous clinical trials to demonstrate safety and efficacy, which can be time-consuming and expensive. The manufacturing of these products involves complex purification and quality control steps, making production costly and susceptible to supply chain disruptions. Furthermore, patent expirations for older generation clotting factors open the door for biosimilar competition, which, while beneficial for affordability, can exert downward pressure on market prices and reduce the revenue potential for innovator companies, influencing their investment in future R&D.
| Restraints | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| High Cost of Clotting Factor Therapies | -1.2% to -1.8% | Global, especially Low & Middle-Income Countries | 2025-2033 (Long-term) |
| Risk of Inhibitor Development in Patients | -0.7% to -1.0% | Global, Clinical Impact | 2025-2033 (Long-term) |
| Stringent Regulatory Landscape and Manufacturing Complexities | -0.5% to -0.8% | Global, particularly Highly Regulated Markets | 2025-2030 (Mid-term) |
| Potential Competition from Biosimilars and Non-Factor Therapies | -0.4% to -0.6% | Developed Markets (North America, Europe) | 2028-2033 (Long-term) |
Clotting Factor Market Opportunities Analysis
The advent and ongoing advancements in gene therapy for bleeding disorders represent the most significant long-term opportunity in the Clotting Factor market. Gene therapy aims to provide a functional copy of the deficient clotting factor gene, potentially offering a one-time, curative treatment for patients, particularly those with hemophilia. While still in its nascent stages for widespread clinical adoption, successful gene therapies could revolutionize patient management by eliminating the need for regular infusions and preventing bleeding episodes. This paradigm shift holds immense potential to improve patient lives dramatically and reshape the entire market, though initial high costs and long-term efficacy remain key considerations. The regulatory approvals of several gene therapies are expected to open up a new era of treatment for severe forms of hemophilia.
The development of novel non-factor therapies and extended half-life (EHL) factors beyond current offerings presents another substantial opportunity. Research is actively exploring treatments that do not rely on direct factor replacement but rather on rebalancing the coagulation cascade, such as monoclonal antibodies (e.g., Emicizumab for hemophilia A with inhibitors) or RNA interference therapeutics. These innovative approaches offer alternative mechanisms of action, potentially addressing limitations of traditional factor replacement, such as inhibitor development or the burden of frequent infusions. Concurrently, ongoing innovation in EHL technologies continues to push the boundaries of less frequent dosing, which remains a key driver for patient and physician preference, ensuring sustained demand for these next-generation products.
The vast untapped potential in emerging markets offers a significant geographical opportunity for market expansion. While North America and Europe currently dominate the Clotting Factor market, regions such as Asia Pacific, Latin America, and the Middle East & Africa have a large undiagnosed or undertreated patient population due to limited access to healthcare, lack of awareness, and economic constraints. As healthcare infrastructure improves, economic conditions strengthen, and diagnostic capabilities expand in these regions, there will be a substantial increase in demand for clotting factor products. Pharmaceutical companies that strategically invest in these markets, address local accessibility challenges, and build educational programs stand to gain significant market share and contribute to global health equity.
| Opportunities | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Emergence of Gene Therapy for Hemophilia | +1.5% to +2.5% | Global, starting with Developed Markets | 2028-2033 (Long-term, transformative) |
| Development of Novel Non-Factor Therapies and Next-Gen EHL Factors | +1.0% to +1.8% | Global, particularly Developed Markets | 2025-2033 (Mid to Long-term) |
| Untapped Potential in Emerging Markets | +0.8% to +1.5% | Asia Pacific, Latin America, MEA | 2025-2033 (Long-term) |
| Orphan Drug Designations and Therapies for Rare Bleeding Disorders | +0.5% to +0.8% | Global, niche markets | 2025-2033 (Long-term) |
Clotting Factor Market Challenges Impact Analysis
One of the primary challenges confronting the Clotting Factor market is the complex and highly specialized nature of manufacturing these biologic products. The production of recombinant clotting factors involves intricate cell culture processes, purification, and quality control measures to ensure product safety, efficacy, and consistency. Any deviation in these processes can lead to supply chain disruptions, product recalls, or delays in new product launches. Furthermore, scaling up production to meet global demand while maintaining stringent quality standards is a continuous challenge. This complexity contributes significantly to the high cost of goods, indirectly impacting market accessibility and affordability, especially in regions with burgeoning demand but limited financial resources.
Ensuring equitable access to clotting factor therapies, particularly in low and middle-income countries (LMICs), remains a significant global health challenge. Despite the rising prevalence of bleeding disorders worldwide, a substantial proportion of patients, especially in LMICs, lack access to diagnosis and appropriate treatment due to economic barriers, limited healthcare infrastructure, and lack of awareness among healthcare professionals. The high cost of therapies further exacerbates this disparity. Addressing this challenge requires multifaceted approaches, including international collaborations, humanitarian aid programs, local manufacturing initiatives, and advocacy for increased government funding and improved reimbursement policies. Without broader access, a significant portion of the global patient population will continue to suffer from preventable bleeding episodes and associated morbidities.
The emergence of novel gene therapies and next-generation non-factor therapies, while presenting significant opportunities, also poses a strategic challenge for traditional clotting factor manufacturers. While these innovations offer curative potential or superior dosing convenience, they could potentially disrupt the long-established market for prophylactic factor replacement. Companies heavily invested in conventional factor products face the imperative to adapt their portfolios, either through internal R&D in these new modalities or through strategic acquisitions and partnerships. Navigating the transition to a landscape potentially dominated by one-time treatments or less frequent injections will require significant shifts in business models, pricing strategies, and patient management approaches, impacting the long-term sustainability of existing product lines. Furthermore, patient adherence to lifelong prophylactic treatment and the burden of frequent infusions remain a clinical challenge, pushing for continuous innovation in administration and product convenience.
| Challenges | (~) Impact on CAGR % Forecast | Regional/Country Relevance | Impact Time Period |
|---|---|---|---|
| Complex Manufacturing and Supply Chain Vulnerabilities | -0.6% to -1.0% | Global | 2025-2033 (Long-term) |
| Limited Access and Affordability in Developing Regions | -0.8% to -1.3% | Emerging Economies (APAC, LATAM, MEA) | 2025-2033 (Long-term) |
| Market Disruption from Gene and Non-Factor Therapies | -0.5% to -0.9% | Global, particularly Developed Markets | 2028-2033 (Long-term) |
| Patient Adherence and Burden of Frequent Infusions | -0.3% to -0.5% | Global, Clinical Practice | 2025-2033 (Long-term) |
Clotting Factor Market - Updated Report Scope
This comprehensive market research report provides an in-depth analysis of the global Clotting Factor market, offering a detailed understanding of its size, growth trajectory, key trends, drivers, restraints, opportunities, and challenges. The report segments the market by product type, application, and region, providing granular insights into market dynamics across various categories and geographical areas. It includes a competitive landscape analysis, profiling key market players, their strategic initiatives, and market share. The report aims to equip stakeholders with actionable intelligence for strategic decision-making, investment planning, and market penetration strategies within the evolving clotting factor therapeutic landscape.
| Report Attributes | Report Details |
|---|---|
| Base Year | 2024 |
| Historical Year | 2019 to 2023 |
| Forecast Year | 2025 - 2033 |
| Market Size in 2025 | USD 15.2 Billion |
| Market Forecast in 2033 | USD 27.9 Billion |
| Growth Rate | 7.8% |
| Number of Pages | 247 |
| Key Trends |
|
| Segments Covered |
|
| Key Companies Covered | Bayer AG, CSL Behring, F. Hoffmann-La Roche Ltd, Grifols S.A., Novo Nordisk A/S, Pfizer Inc., Sanofi, Shire (now Takeda Pharmaceutical Company Limited), BioMarin Pharmaceutical Inc., Octapharma AG, Kedrion Biopharma, Spark Therapeutics (acquired by Roche), uniQure N.V., Genentech Inc., Amgen Inc., Emergent BioSolutions, Biomarin Pharmaceutical Inc., Chiesi Farmaceutici S.p.A., Alnylam Pharmaceuticals, Sangamo Therapeutics |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Speak to Analyst | Avail customised purchase options to meet your exact research needs. Request For Analyst Or Customization |
Segmentation Analysis
The Clotting Factor market is broadly segmented by product type, application, distribution channel, and end-use, providing a granular view of market dynamics and growth opportunities across various categories. The product type segment differentiates between recombinant clotting factors, which dominate due to their safety profile and continuous innovation (e.g., extended half-life variants), and plasma-derived clotting factors, still vital for certain indications and patient populations. Within these, specific factors like Factor VIII, Factor IX, and Factor VIIa are critical, addressing different types of hemophilia and bleeding disorders. Bypassing agents form a distinct subsegment, crucial for managing patients who develop inhibitors to standard factor replacement therapy.
By application, the market is primarily driven by the treatment of Hemophilia A and Hemophilia B, which represent the most prevalent inherited bleeding disorders requiring lifelong therapy. However, the market also includes treatments for Von Willebrand Disease and other rare factor deficiencies, highlighting the expanding scope of therapeutic interventions. The distribution channel segment typically covers hospital pharmacies, retail pharmacies, and the growing segment of online pharmacies, reflecting the varied patient access points. Finally, the end-use segment identifies where these treatments are primarily administered or utilized, with hospitals and homecare settings being dominant due to the nature of the therapy.
Each segment presents unique growth drivers and challenges. For instance, the recombinant factor segment is propelled by R&D advancements and patient preference for safer, more convenient options, while plasma-derived factors maintain their niche due to established efficacy and specific indications. The homecare setting is rapidly gaining traction due to the convenience it offers for chronic disease management, reducing hospital visits and improving patient autonomy. Understanding these segment-specific dynamics is crucial for market participants to tailor their strategies and product offerings effectively.
- By Product Type:
- Recombinant Clotting Factors (Factor VIII, Factor IX, Factor VIIa, Von Willebrand Factor)
- Plasma-Derived Clotting Factors (Factor VIII, Factor IX, Factor VIIa, Von Willebrand Factor, Prothrombin Complex Concentrates (PCCs))
- Bypassing Agents
- By Application:
- Hemophilia A
- Hemophilia B
- Von Willebrand Disease
- Other Factor Deficiencies (e.g., Factor VII, X, XIII deficiencies)
- Acquired Hemophilia
- By Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- By End-Use:
- Hospitals
- Clinics
- Homecare Settings
Regional Highlights
- North America: This region dominates the Clotting Factor market, primarily due to the high prevalence of bleeding disorders, advanced healthcare infrastructure, significant R&D investments, and favorable reimbursement policies. The presence of key market players and early adoption of novel therapies, including extended half-life factors and gene therapies, further solidify its leading position. The United States accounts for the largest share within North America, driven by high per-patient treatment costs and a well-established patient support system.
- Europe: Europe represents another significant market share due to its well-developed healthcare systems, high awareness of bleeding disorders, and robust government support for rare disease treatments. Countries like Germany, France, and the UK are major contributors, benefiting from strong research activities and a high rate of diagnosis and prophylactic treatment. However, pricing pressures and heterogeneous reimbursement policies across different European countries present some challenges.
- Asia Pacific (APAC): The APAC region is poised for the highest growth rate during the forecast period. This growth is attributable to an increasing patient pool, improving healthcare expenditure, rising awareness about bleeding disorders, and the developing healthcare infrastructure in countries such as China, India, and Japan. While access to advanced therapies remains a challenge in some parts, the expanding middle class and efforts by governments and non-profit organizations to improve diagnosis and treatment are driving market expansion.
- Latin America: This region is an emerging market with significant growth potential, albeit from a smaller base. Brazil and Mexico are leading contributors, characterized by increasing healthcare investments and efforts to improve access to diagnosis and treatment for hemophilia. Economic challenges and varying healthcare policies, however, influence the pace of market development.
- Middle East and Africa (MEA): The MEA region is expected to show steady growth. Gulf Cooperation Council (GCC) countries lead the adoption of advanced therapies due to high per capita income and robust healthcare spending. However, challenges persist in other parts of Africa due to limited healthcare resources, low awareness, and diagnosis rates, creating substantial unmet needs but also long-term growth opportunities as infrastructure improves.
Top Key Players
The market research report includes a detailed profile of leading stakeholders in the Clotting Factor Market.- Bayer AG
- CSL Behring
- F. Hoffmann-La Roche Ltd
- Grifols S.A.
- Novo Nordisk A/S
- Pfizer Inc.
- Sanofi
- Shire (now Takeda Pharmaceutical Company Limited)
- BioMarin Pharmaceutical Inc.
- Octapharma AG
- Kedrion Biopharma
- Spark Therapeutics (acquired by Roche)
- uniQure N.V.
- Genentech Inc.
- Amgen Inc.
- Emergent BioSolutions
- Chiesi Farmaceutici S.p.A.
- Alnylam Pharmaceuticals
- Sangamo Therapeutics
- CSL Ltd.
Frequently Asked Questions
What are clotting factors and why are they important?
Clotting factors are proteins in the blood that work together to form a blood clot, stopping bleeding after an injury. They are crucial for hemostasis, the body's natural process to prevent and stop bleeding. Deficiencies or abnormalities in these factors lead to bleeding disorders like hemophilia, where even minor injuries can cause prolonged and dangerous bleeding episodes. Replacement therapy with concentrated clotting factors is the standard treatment to manage these conditions and prevent life-threatening hemorrhages.
What are the primary types of hemophilia treated with clotting factors?
The primary types of hemophilia treated with clotting factors are Hemophilia A and Hemophilia B. Hemophilia A results from a deficiency in clotting Factor VIII, accounting for about 80% of all hemophilia cases. Hemophilia B is caused by a deficiency in clotting Factor IX. Both are X-linked genetic disorders predominantly affecting males. Treatment involves regular infusions of the deficient clotting factor to prevent bleeding (prophylaxis) or to stop bleeding episodes (on-demand treatment).
How has treatment for bleeding disorders evolved over time?
Treatment for bleeding disorders has evolved significantly. Initially, plasma-derived clotting factors carried risks of viral transmission. The advent of recombinant DNA technology revolutionized treatment by producing highly pure, virus-free clotting factors. More recently, extended half-life (EHL) factors have emerged, reducing injection frequency and improving patient convenience. The frontier of treatment now includes gene therapy, offering the potential for a one-time functional cure by enabling the body to produce its own clotting factors, and non-factor therapies that rebalance the coagulation cascade.
What are the main challenges facing the Clotting Factor market?
Key challenges in the Clotting Factor market include the high cost of therapies, which limits access in many regions, especially low and middle-income countries. The risk of inhibitor development in patients, where the immune system attacks the infused factor, complicates treatment significantly. Manufacturing complexities for these specialized biologics can lead to supply chain vulnerabilities. Additionally, ensuring equitable global access and managing the disruptive potential of emerging gene and non-factor therapies are ongoing challenges.
What is the future outlook for the Clotting Factor market?
The future outlook for the Clotting Factor market is robust and dynamic. It is expected to continue growing, driven by advancements in gene therapy and novel non-factor therapies that promise more convenient and potentially curative options. Extended half-life factors will remain significant due to their improved convenience. While cost and access remain challenges, increasing awareness, improved diagnostics, and expansion into emerging markets will contribute to sustained demand. The market is shifting towards more personalized and patient-centric treatment approaches, with innovation focused on enhancing patient quality of life and potentially offering long-term solutions for bleeding disorders.